International Journal of Nanomaterials, Nanotechnology and Nanomedicine
Preparation of nanoxylan from wheat straw
School of Chemistry and Chemical Engineering, Northwestern Polytechnical University, Xi’an, 710129, Shaanxi, PR China
Author and article information
Cite this as
Sheraz M, Sun XF, Siddiqui A. Preparation of nanoxylan from wheat straw. Int J Nanomater Nanotechnol Nanomed. 2024; 10(2): 88-91. Available from: 10.17352/2455-3492.000067
Copyright License
© 2024 Sheraz M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.In the present study, attention has been focused on the preparation of nanoxylan from wheat straw, and a simple and scale-up method for the synthesis of high-purity nanoxylan from wheat straw was presented. This preparation method was performed in a series of steps including alkaline peroxide extraction, ethanol precipitation, and freeze-drying. These steps ensure high purity and yield of nanoxylan under controllable and cost-effective conditions. The particle diameters of the prepared nanoxylan ranged from 30 nm to 90 nm with more than 90% xylose content and a weight-average molecular weight of 15,000–30,000 g/mol. Such unique properties as high dispersibility and solubility provide nanoxylan with huge potential applications in food packaging, biomedicine, agriculture, and so on.
Xylan is a polymeric sugar and one major hemicellulose component present throughout plant biomass and in much greater amounts in certain feedstocks such as straws, grasses, and hardwoods [1-5]. Xylan accounts for 20% - 35% of wood fiber, although its detailed structure varies between species and plant tissues [6]. Through the side chains, different xylan derivatives are produced from β-D-1,4-xyloside bonds in a linear main chain: glucuronoxylan [7], arabinoxylan [8] and glucurono-arabinoxylan. This unique molecular structure confers remarkable biological activity on xylan, as it manifests excellent physiological functions [9,10]. Concerning cellulose and chitosan, xylan demonstrates excellent hydrophilicity, biocompatibility, biodegradation characteristics, and even antitumor properties [11-13]. These attributes make it highly valued in the food industry for its incorporation into functional foods used in the management of hyperglycemia, and hyperlipidemia [14]. Besides, xylan is used in dairy products, beverages, and edible films, enhancing taste, nutrient absorption, and environmental sustainability [15].
In recent years, great interest has been focused on the conversion of xylan into nanoparticles, the so-called nanoxylan. The mechanical properties, thermal stability, and barrier properties of nanoxylan have been greatly improved with nanoscale dimensions. These improvements widen its application area in food packaging, biomedicine, and tissue engineering [16-18]. For example, nanoxylan-based materials have shown excellent drug-loading capacity with controlled and targeted drug delivery and few side effects [19,20]. The functional films from nanoxylan were safe, biodegradable, and energy-efficient, and the preparation met the requirement for sustainable development. However, poor dispersibility and solubility seriously limit the practical utility of the traditional xylan particles [21,22]. Improvements in the mechanical and barrier properties are similarly presented in the nanoparticles discussed in the paper; moreover, the nanoscale benefits of nanoxylan open bright prospects in those fields. Besides, their compatibility with sustainable development goals speaks for their application in food packaging, medicine, and similar industries. Further research aimed at improving their dispersibility and solubility could probably enable the extension of their practical applications.
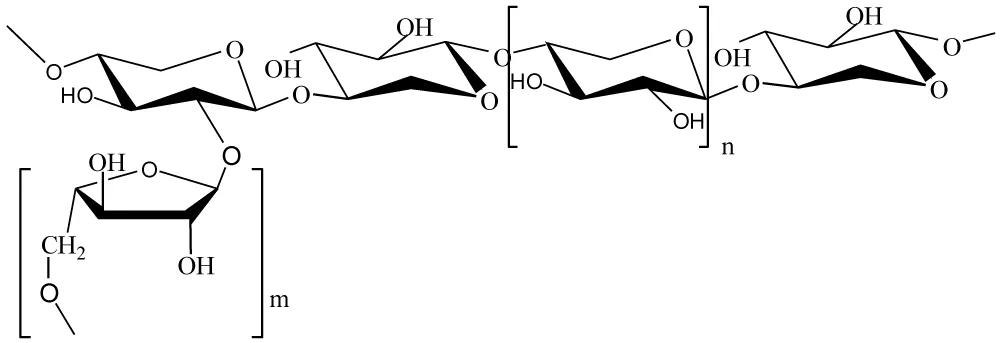
This study focuses on how xylan is being redesigned into nanoxylan to avoid the problems of using regular xylan, given its poor dispersibility and solubility. Nanoxylan shows much better mechanical properties, thermal stability, and barrier performance; thus, it is fitting for usage in food packaging, biomedicine, and even tissue engineering. This work presents a significant enhancement of functionality, employing advanced synthesis and characterization techniques while being aligned with the goals of sustainable development. In this paper, a simple scale-up method for the synthesis of high-purity nanoxylan from wheat straw was presented. The method proposed here comprised a series of well-controlled steps, such as alkaline peroxide extraction, ethanol precipitation, and freeze-drying. Here the particle diameter of nanoxylan was 30-90 nm, and the structural formula of xylan is as follows (Figure 1).
Experimental
Materials
Wheat straw powder, the primary raw material, was collected from fields in Xi’an, China. Reagents, including NaOH, EDTA, H₂O₂, NaBH₄, and ethanol, were of analytical grade and obtained from standard suppliers (Tianjin, China). Deionized water was used in all preparation steps.
Extraction of xylan from wheat straw
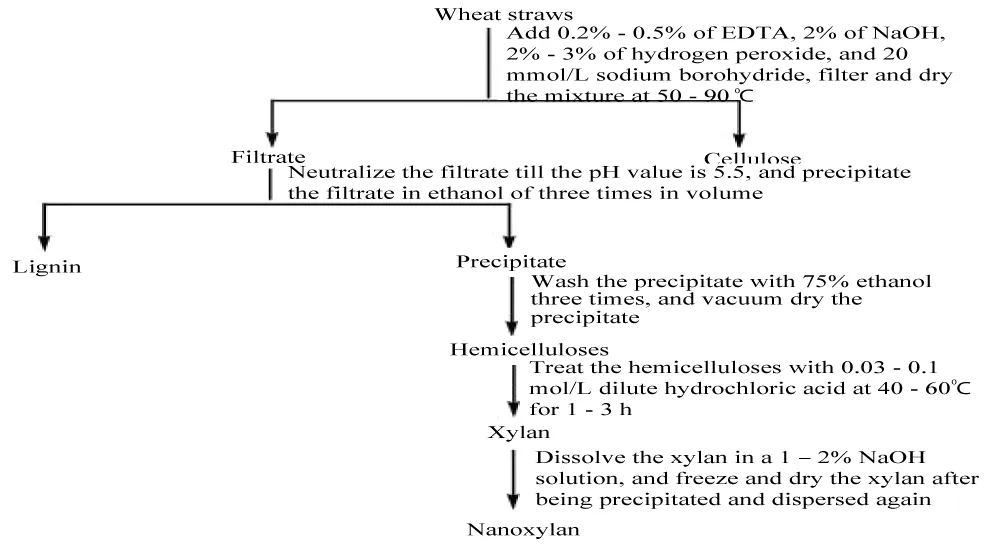
First, wheat straws were crushed and passed through a 40-mesh sieve. In method I, 30 g of the wheat straw powder was dissolved in 750 mL of water, and 3.75 g of EDTA was added. The mixture was reacted at 80 °C in a constant temperature oil bath for 1 hour, then filtered and washed. After drying, the sample was mixed with 600 mL of water, 12.25 g of NaOH, and 63.7 mL of a 30% H₂O₂ solution, and the reaction was carried out at 90 °C for 2 hours. Following this, 20 mmol/L NaBH₄ was added for a 4-hour reaction. The solution, upon cooling, was filtered, and concentrated, and three times its volume of ethanol was added to the filtrate to precipitate the xylan. It was then filtered and dried at 60 °C. In method II, the process was similar, with the NaOH amount being 12.25 g, that of H₂O₂ being 54.6 ml, and the reaction temperature reduced to 80 °C. In the case of method III, the mixture consists of 2.25 g EDTA, the reaction temperature at 90 °C, and 42.8 mL of 30% H₂O₂. On the other hand, a mixture consisting of 2.1 g of EDTA and 31.58 mL of H₂O₂ was used for method IV at 50 °C reaction temperature. After the reactions, subsequent hemicellulose treatments with 0.1 mol/L hydrochloric acid at 40 °C -60 °C were used for the provision of xylan (Figure 2).
Preparation of nanoxylan
To prepare nanoxylan, the extracted xylan was dissolved in NaOH solution and neutralized with dilute acid to pH 5.5, and then precipitated by ethanol. The filter cake, after filtration, was re-dissolved in ethanol and freeze-dried to get nanoxylan. In method I, 1 g of xylan was dissolved in 2% NaOH (50 mL) at 60 °C, neutralized, precipitated, and dried. Method II follows the same procedures as described above except that 100 mL of 2% NaOH was used at 50 °C. The xylan, according to method III, was dissolved in 200 mL of NaOH at a temperature of 40 °C, while according to method IV, it was dissolved in 200 mL of NaOH at 30 °C, followed by precipitation and drying steps to obtain the nanoxylan.
Results and discussion
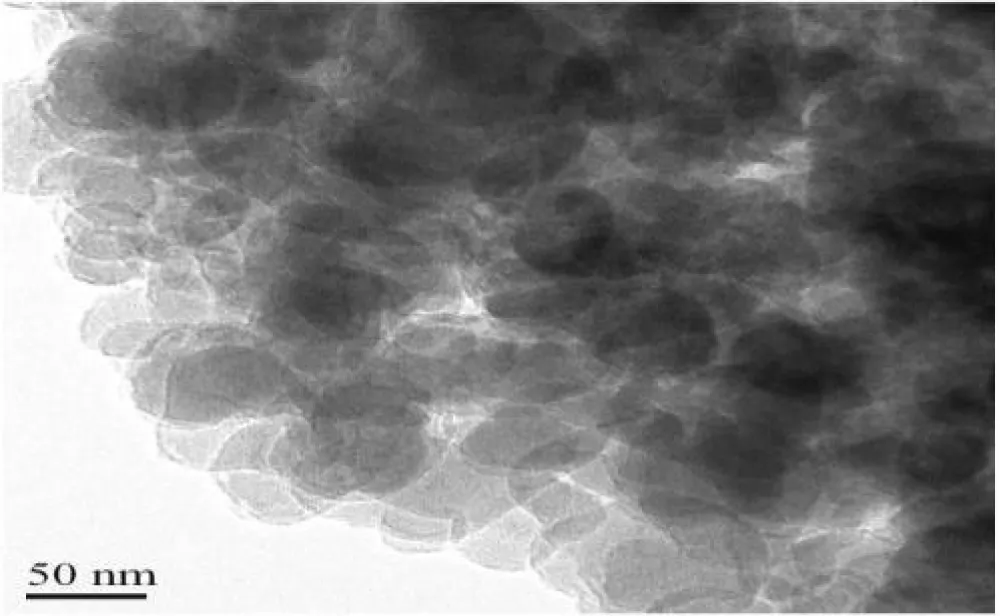
In Figure 3, TEM analysis shows the morphology and size distribution of nanoxylan particles. The nanoparticles are spherical in structure, with sizes on the same scale, ranging from 30 nm to 90 nm. As evidenced, nanoscale dimensions are important to improve the dispersibility, solubility, and functional properties of xylan. There is no evidence of any significant aggregation, which means the preparation method prevents the clumping of the particles, hence producing a quality product. Figure 3 points out that the particle size distribution of nanoxylan is within the range of 30-90 nm but in spherical shapes uniformly. Poor aggregation indicates that the preparation method was quite effective in giving a well-dispersed particulate system. An appropriately controlled size distribution can improve the dispersibility, solubility, and functionality of nanoxylan for various applications [22].
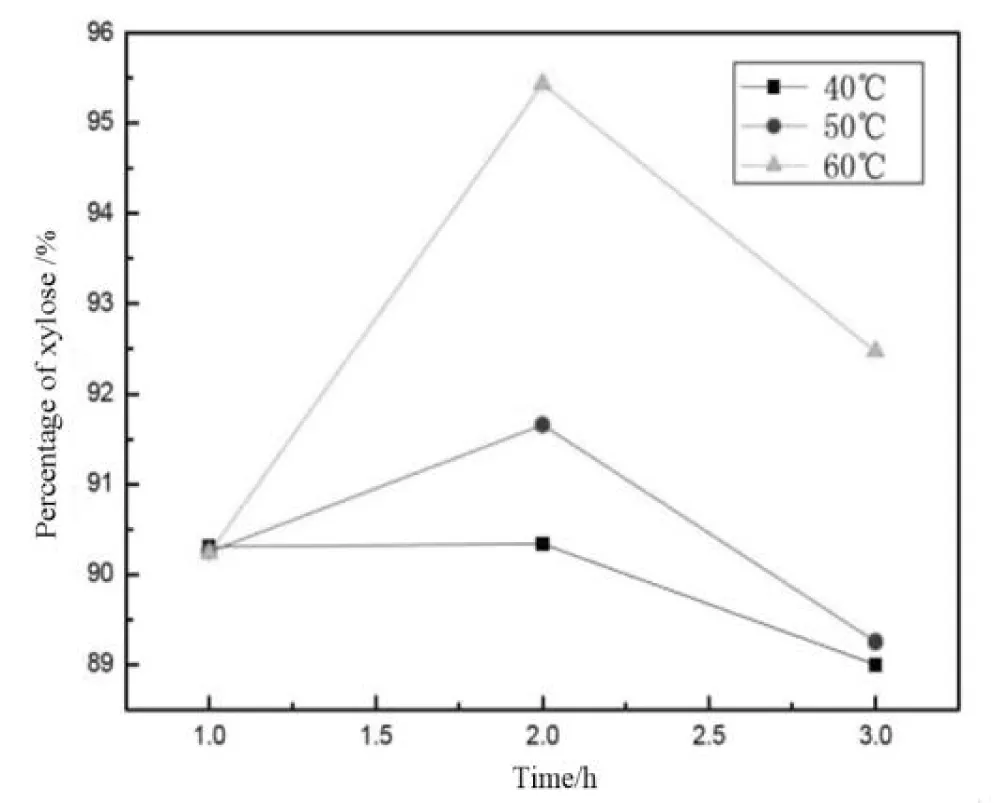
Table 1 shows the molecular weights of four nanoxylan samples, and Table 2 shows the monosaccharide composition of four nanoxylan samples. The treatment temperature of dilute hydrochloric acid had a significant influence on the molecular weights and the xylose percent of nano xylans, and the molecular weight of prepared nano xylan decreased and the xylose percent increased with increasing temperature [24,25]. Figure 4 shows the dependence of treatment time and temperature of \dilute hydrochloric acid versus the xylose content in xylan. This graph indeed shows that with an increase in the treatment time and/or raising the temperature to a certain value within the working range, one observes a positive trend in the increasing content of xylose. Fairly higher temperatures, for example, 60 °C, succeed in accelerating the rate of hydrolysis by increasing the release of xylose from hemicelluloses. On the other hand, in very high temperatures, though, degradation also may surpass the optimum, as revealed in the graph. With each increase in the time of treatments, the content increases steadily as the treatment goes up from 0 to 3, which postulates that sufficient duration of reaction time is one of the most critical factors in optimal xylose release. The figure identifies an optimal combination that maximizes moderate to highly elevated temperatures (40 °C - 60 °C) and treatment duration (1–3 hours) while minimizing degradation or loss. The reduction of xylan percentage after 2 hours can be attributed to the degradation of xylose, the main hydrolysis product, into products such as furfural and formic acid during prolonged acid treatment. This emphasizes the kinetic interplay involving hydrolysis and degradation hence underscoring the need for optimizing the treatment time to achieve maximum yields in xylose while minimizing losses [26,27].
Conclusions and future perspectives
In this work, a scalable method for preparing nanoxylan from wheat straw with high purity and nanoscale particle sizes (30 nm - 90 nm) was developed. Optimal conditions for diluting hydrochloric acid hydrolysis, such as 40 °C - 60 °C for 1-3 hours, were obtained to maximize the xylose content of nanoxylan for efficient extraction and processing. The resulting nanoxylan can be applied in biodegradable packaging and targeted drug delivery. More optimization industrially for scaling regarding the environment needs to be done. Nevertheless, this work hereby places nano xylan among promising biomaterials in various applications through which sustainability is combined with advances in functionality. In the future, research should focus on improving the dispersibility and solubility of nanoxylan in wider applications. Making nanoxylan compatible with various materials such as biopolymers and drug carriers merits adding exploration to this aspect of research. Other important contributions will include the environmental impact of nano xylan production as well as life cycle assessments because such evaluations will ensure that the commercialization of the product is sustainable and eco-friendly at the same time. The efforts will also focus on ever more collaboration between academia and industry towards developing cost-effective methods of scaling up production without compromising the quality and performance of the product. Such breakthroughs should realize nanoxylan into active food packaging, drug delivery systems, or state-of-the-art biomaterials for tissue engineering applications.
This research was funded by the Key R&D project of Shaanxi Province (No. 2023-YBNY-261) and the Xi’an Science and Technology Plan Project (No. 2024JH-NYYB-0146).
- Dafchahi MN, Acharya B. Green extraction of xylan hemicellulose from wheat straw. Biomass Conversion and Biorefinery. 2024;14(17):21229-21243. Available from: http://dx.doi.org/10.1007/s13399-023-04383-7
- Nongkhai SN. Hydrolysis of grass xylan by xylanases from Aureobasidium pullulans for xylooligosaccharide and xylitol productions. 2022. Available from: https://digital.car.chula.ac.th/chulaetd/5747/
- Lehuedé L, Henríquez C, Carú C, Córdova A, Mendonça RT, Salazar O. Xylan extraction from hardwoods by alkaline pretreatment for xylooligosaccharide production: A detailed fractionation analysis. Carbohydrate Polymers. 2023;302:120381. Available from: https://doi.org/10.1016/j.carbpol.2022.120381
- Ngo T-D, Chandra R, Ahvazi B. Sustainable lignocellulosic nanomaterials for future green applications. In: Handbook of Nanomaterials and Nanocomposites for Energy and Environmental Applications. Springer; 2021. p. 3445-3464. Available from: https://link.springer.com/referenceworkentry/10.1007/978-3-030-36268-3_19
- Khaire KC, Sharma K, Thakur A, Moholkar VS, Goyal A. Extraction and characterization of xylan from sugarcane tops as a potential commercial substrate. J Biosci Bioeng. 2021;131(6):647-654. Available from: https://doi.org/10.1016/j.jbiosc.2021.01.009
- Silva TAL, Varão LHR, Pasquini D. Lignocellulosic Biomass. In: Handbook of Biomass. Springer; 2024. p. 105-142. Available from: https://link.springer.com/referenceworkentry/10.1007/978-981-99-6727-8_5
- Sivan P, Heinonen E, Escudero L, Gandla ML, Jiménez-Quero A, Jönsson LJ, et al. Unraveling the unique structural motifs of glucuronoxylan from hybrid aspen wood. Carbohydr Polym. 2024 Nov 1;343:122434. Available from: https://doi.org/10.1016/j.carbpol.2024.122434
- Wang J, Bai J, Fan M, Li T, Li Y, Qian H, et al. Cereal-derived arabinoxylans: Structural features and structure-activity correlations. Trends Food Sci Technol. 2020;96:157-165. Available from: http://dx.doi.org/10.1016/j.tifs.2019.12.016
- Wang H, Yang H, Wen Z, Gao C, Gao Y, Tian Y, et al. Xylan-based nanocompartments orchestrate plant vessel wall patterning. Nat Plants. 2022;8(3):295-306. Available from: https://doi.org/10.1038/s41477-022-01113-1
- Fu GQ, Zhang SC, Chen GG, Hao X, Bian J, Peng F. Xylan-based hydrogels for potential skin care application. Int J Biol Macromol. 2020 Apr 30;158:244-250. Available from: https://doi.org/10.1016/j.ijbiomac.2020.04.235
- Zhao B, Li H, Tian K, Su Y, Zou Z. Synthesis and antitumor activity of bagasse xylan derivatives modified by graft-esterification and cross-linking. Int J Biol Macromol. 2023;253(Pt 3):126867. Available from: https://doi.org/10.1016/j.ijbiomac.2023.126867
- Qin Y, Peng X. Synthesis of biocompatible cholesteryl–carboxymethyl xylan micelles for tumor-targeting intracellular DOX delivery. ACS Biomater Sci Eng. 2020;6(3):1582-1589. Available from: https://doi.org/10.1021/acsbiomaterials.0c00090
- Khaire KC, Maibam PD, Thakur A, Goyal A. Biomedical and pharmaceutical applications of xylan and its derivatives. In: Hemicellulose Biorefinery: A Sustainable Solution for Value Addition to Bio-Based Products and Bioenergy. Springer; 2022;447-465. Available from: https://link.springer.com/chapter/10.1007/978-981-16-3682-0_14
- Chakraborty M, Bhowal J. Prospects of health beneficial functional xylooligosaccharides produced by enzymatic hydrolysis of xylan and application in food industry: A comprehensive review. Food Rev Int. 2024:1-28. Available from: https://doi.org/10.1080/87559129.2024.2412617
- Nechita P, Mirela R, Ciolacu F. Xylan hemicellulose: A renewable material with potential properties for food packaging applications. Sustainability. 2021;13(24):13504. Available from: https://doi.org/10.3390/su132413504
- Oliveira I, Filho D, Queirós M, Santos C, Miranda J, Arruda P, et al. Xylans as a promising prebiotic agent: A brief review. Sci Electron Arch. 2024;17. Available from: https://doi.org/10.36560/17320241910
- Nechita P, Roman M, Năstac SM. Green approaches on modification of xylan hemicellulose to enhance the functional properties for food packaging materials—A review. Polymers. 2023;15(9):2088. Available from: https://doi.org/10.3390/polym15092088
- Wijaya CJ, Ismadji S, Gunawan S. A review of lignocellulosic-derived nanoparticles for drug delivery applications: Lignin nanoparticles, xylan nanoparticles, and cellulose nanocrystals. Molecules. 2021;26(3):676. Available from: https://doi.org/10.3390/molecules26030676
- Long J, Zhou G, Yu X, Xu J, Hu L, Pranovich A, et al. Harnessing chemical functionality of xylan hemicellulose towards carbohydrate polymer-based pH/magnetic dual-responsive nanocomposite hydrogel for drug delivery. Carbohydr Polym. 2024;343:122461. Available from: https://doi.org/10.1016/j.carbpol.2024.122461
- Fan S, Gao X, Pang J, Liu G, Li X. Enhanced preservative performance of pine wood through nano-xylan treatment assisted by high-temperature steam and vacuum impregnation. Materials. 2023;16(11):3976. Available from: https://doi.org/10.3390/ma16113976
- Macedo JVC, Abe MM, Sanvezzo PB, Grillo R, Branciforti MC, Brienzo M. Xylan-starch-based bioplastic formulation and xylan influence on the physicochemical and biodegradability properties. Polymer Bull. 2023;80(7):8067-8092. Available from: https://link.springer.com/article/10.1007/s00289-022-04385-x
- Gao X, Fan S, Pang J, Rahman MZ, Zhu D, Guo S, et al. Preparation of nano-xylan and its influences on the anti-fungi performance of straw fiber/HDPE composite. Ind Crops Prod. 2021;171:113954. Available from: https://doi.org/10.1016/j.indcrop.2021.113954
- Wang J, Minami E, Kawamoto H. Thermal reactivity of hemicellulose and cellulose in cedar and beech wood cell walls. J Wood Sci. 2020;66:1-10. Available from: https://link.springer.com/article/10.1186/s10086-020-01888-x
- Yuan Q, Liu S, Ma MG, Ji XX, Choi SE, Si C. The kinetics studies on hydrolysis of hemicellulose. Front Chem. 2021;9:781291. Available from: https://doi.org/10.3389/fchem.2021.781291
- Rahmani AM, Tyagi VK, Kazmi AA, Ojha CSP. Hydrothermal and thermal-acid pretreatments of wheat straw: Methane yield, recalcitrant formation, process inhibition, kinetic modeling. Energy. 2023;283:129083. Available from: https://doi.org/10.1016/j.energy.2023.129083
- Torget R, Teh-An H. Two-temperature dilute-acid prehydrolysis of hardwood xylan using a percolation process. Appl Biochem Biotechnol. 1994;45:5-22. Available from: https://link.springer.com/article/10.1007/BF02941784
- Beckendorff A, Lamp A, Kaltschmitt M. Optimization of hydrolysis conditions for xylans and straw hydrolysates by HPLC analysis. Biomass Conv Bioref. 2023;13:3361-3374. Available from: https://link.springer.com/article/10.1007/s13399-021-01429-6
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
