Open Journal of Analytical and Bioanalytical Chemistry
Advancements in Two-Dimensional Liquid Chromatography for Enhanced Food and Environmental Analysis
School of Pharmacy, Lingaya’s Vidyapeeth, Faridabad-121002, Haryana, India
Author and article information
Cite this as
Chopra A. Advancements in Two-Dimensional Liquid Chromatography for Enhanced Food and Environmental Analysis. Open J Anal Bioanal Chem. 2026; 10(1): 1-14. Available from: 10.17352/ojabc.000034
Copyright License
© 2026 Chopra A. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Two-dimensional liquid chromatography (2D-LC) represents a significant advancement in food analysis, addressing the limitations of one-dimensional liquid chromatography (1D-LC) in separating complex food matrices. Food analysis plays a critical role in assessing the components, physicochemical properties, sensory qualities, and contamination risks of food products. Given the rising interest in identifying bioactive compounds in food due to their health benefits, the need for enhanced analytical techniques has become more pressing. Traditional 1D-LC often struggles to resolve closely related and co-eluting species in complex food samples, necessitating methods with greater resolving power. 2D-LC offers a valuable alternative by coupling two independent separation processes, thereby significantly increasing separation efficiency. This approach is particularly effective when paired with mass spectrometry (MS), which enhances detection sensitivity and specificity, allowing for more accurate structural identification. Moreover, 2D-LC has proven instrumental in environmental analysis, particularly in detecting and characterizing organic micropollutants (OMPs) at trace levels. As the demand for comprehensive food analysis and environmental monitoring grows, 2D-LC coupled with MS is poised to play an increasingly vital role in ensuring food safety, quality, and informed consumer choices. Therefore, this review briefly introduces the working principle of 2D-LC concerning its application in the field of food and environmental analysis in recent years.
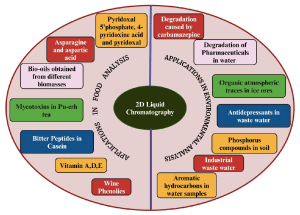
Graphical abstract: Various applications of 2D-LC in food and environmental analysis.
2D-LC; HRMS; 1D-LC; OMP; 2D-LC-MS; RPLC
Introduction
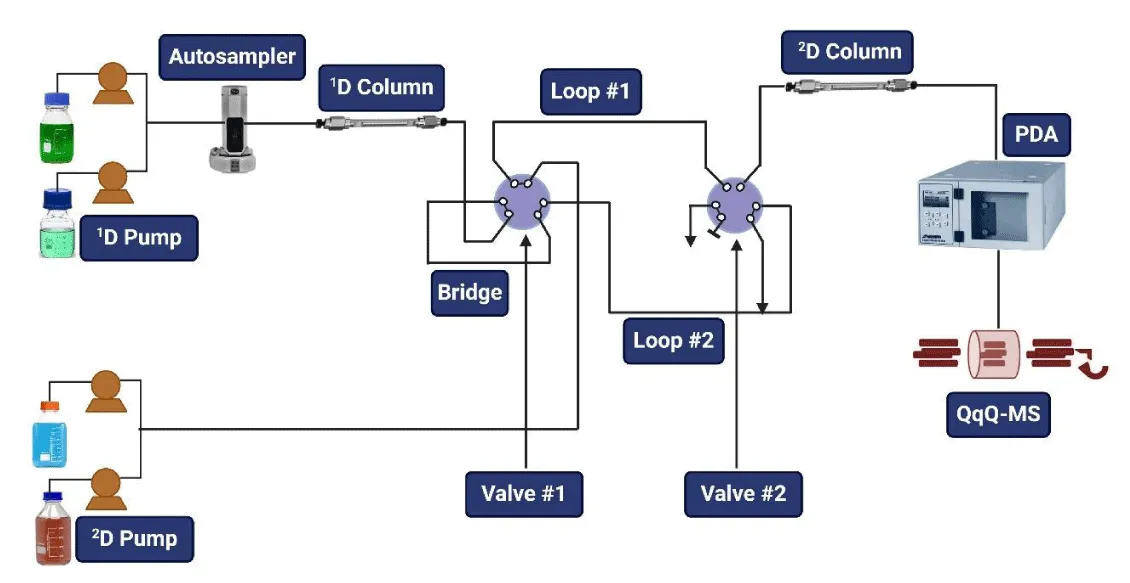
Food analysis confines the study, creation, and use of analytical techniques to describe the components and attributes of food and food-related products. These processes offer vital information on the components found in food as well as its physicochemical characteristics, sensory qualities, and potential for contamination [1]. They are essential for maintaining food quality and safety [2,3]. Furthermore, because these molecules have bioactive roles in plants and the human diet, determining the bioactive content of food and food-based products is receiving a lot of interest [4,5]. The field of food analysis is concerned with the creation, examination, and use of analytical techniques to characterize the characteristics of foods and their ingredients [6]. The capacity to economically produce food that is consistently safe, nutrient-dense, and appealing, as well as the ability for consumers to make educated diet decisions, all depend on our ability to rationally grasp the components that impact food attributes [7]. Liquid chromatography (LC) is considered one of the most powerful tools in terms of analytical separation techniques [8]. Coupling two separate but independent separation processes offers 2D-liquid chromatography (2D-LC) [9]. This results in a large increase in separation power since a sample is analyzed by two distinct separation methods. Gaining a decent resolution of the components present in complex samples that are not separable using one-dimensional procedures requires an increase in separation provided by two-dimensional liquid chromatography (2D-LC) techniques [10]. When mass spectrometry (MS) is used in conjunction with chromatographic separation mechanisms, the various mechanisms’ versatility and selectivity become more significant. This lowers the detection limits for quantitative analysis and, because of the high specificity of MS and sensitivity, yields structural identification [11,12]. However, traditional one-dimensional liquid chromatography (1D-LC) frequently fails to resolve a large number of substances when particularly heterogeneous and complicated food samples need to be analyzed [13]. One such problem that 1D-LC needs to address is the analysis of mixtures including closely related and co-eluting species. These constraints necessitate the use of methods with more resolving power, and two-dimensional liquid chromatography (2D-LC) proves to be a valuable substitute to take into account in this regard [9]. Also, for analytical scientists, analyzing complex environmental matrices is extremely difficult since there are so many different substances, both known and undiscovered, with a wide range of chemical and physical characteristics. The development of efficient instruments to decipher the chemical composition of such environmental samples has been spurred by the necessity for a comprehensive characterization of this complexity [14]. These days, one of the most significant LC-MS application fields is environmental analysis, which is mostly concerned with the investigation of the presence and fate of organic micropollutants (OMPs). The majority of OMPs are polar pollutants that are released into the environment at ppm levels or less during manufacture, use, and disposal. OMPs contain the so-called “emerging contaminants” in addition to traditional pollutants, which are mostly insecticides [15]. The primary principle of 2D-LC is to increase the separation power by analyzing a single sample using two orthogonal separation methods [8,16]. After injecting the sample into the first column, or first dimension (1D), one or more fractions of the 1D effluent are moved to the second dimension (2D) and go through one more separation [17]. This allows for the resolution of compounds in the 2D separation process that were not able to be separated in the 1D retention mechanisms [18, =19]. The high efficiency of separation provided by 2D-LC merged with highly sensitive and selective MS makes 2D-LC-MS one of the most powerful strategies for food analysis as well as environmental analysis [1,10,20]. The methodology used to analyze the entire sample is referred to as “comprehensive” (LC-LC), and it has been known as a novel analytical approach for food and environmental analysis due to its wide variety of applications, as evidenced by the current literature [18, 21, 22]. A vast range of analyzers with various features and capacities has been made possible by recent advancements in MS instrumentation [23,24]. Tandem spectrometry (MS/MS) [25,26] or high-resolution MS (HRMS) [27, 28] are employed in most applications to acquire the most effective identification since they can provide precise masses and dependable structure elucidation by fragmentation. Additionally, separations can be comprehensive (LC-LC) if the entire sample is analyzed via the two dimensions or heart-cutting (LC-LC) if just certain fractions from the 1D are moved to the 2D [29]. The latter scenario should not result in the loss of the separation achieved in the 1D [30,31]. This criterion suggests that a comparatively high sample frequency from 1D effluent should be applied. As a result, compared to traditional 1D-LC, the use of two dimensions greatly improves and boosts the separation power property that may be very helpful in studies about food analysis. Figure 1 depicts the overall structure of 2D-LC. Additionally, this technology may be coupled with mass spectrometry, as with other liquid chromatography-based technologies, to further enhance separation and identification capabilities. Readers who are interested in the practical elements of 2D-LC are directed to several well-published and outstanding evaluations [6,18,19,32]. The goal of this review article is to familiarize food analytical chemists with the general elements of the LC x LC technique. This is followed by a part that focuses on the practical application of the LC x LC technique, which includes the design of the newest interfaces and will be especially interesting to advanced readers.
2D Liquid chromatography layout
Different varieties of 2D-LC can be distinguished based on multiple criteria: (i) it is feasible to distinguish between online mode, offline mode, and a stop-and-go intermediate system based on how the wastewater from the 1D is transported to the 2D. (ii) 2D-LC can be split into heart-cutting (LC-LC) and comprehensive (LC × LC) chromatography depending on how many fractions from the 1D effluent are moved to the 2D column. This section presents an explanation of the various methods, along with the potential connection between the two dimensions.
Different offline, online, and stop-and-go variations
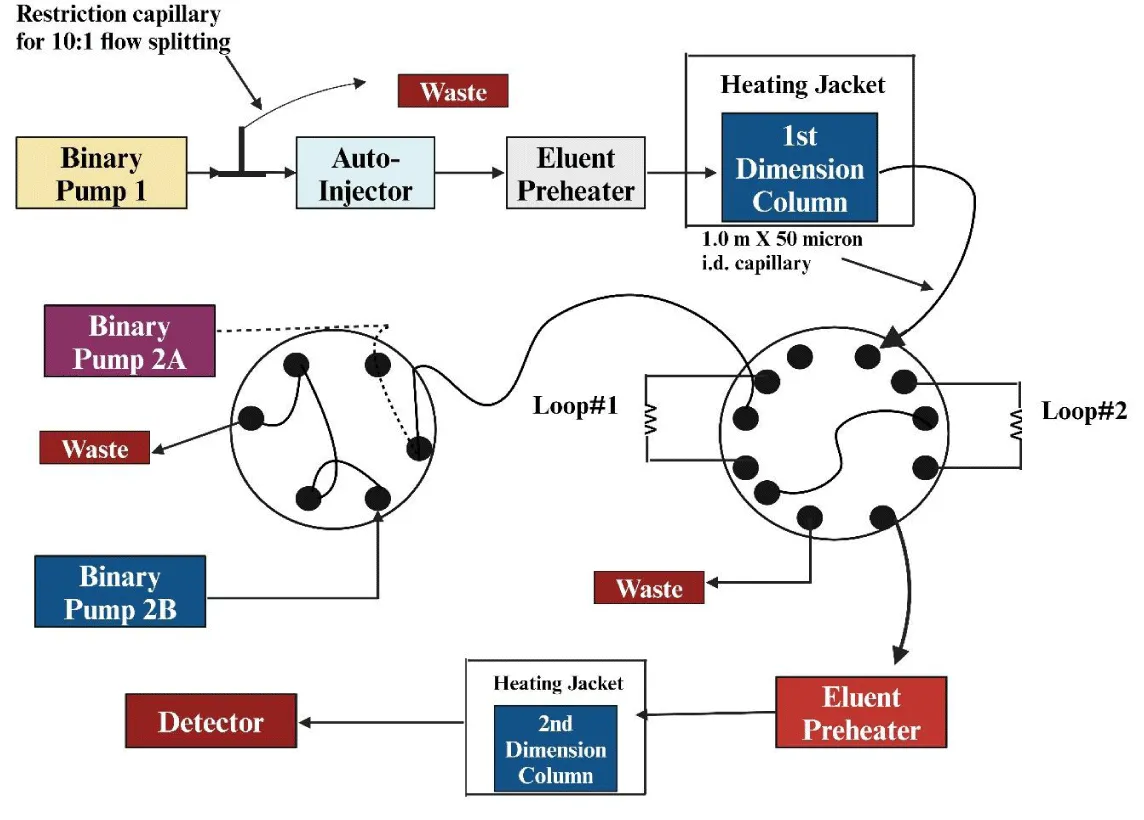
Fractions from the 1D drainage are manually collected and fed into the 2D in offline separations. The benefits of this 2D approach include the potential for slower separations in the 2D, easy instrumentation, and sample modification between analyses in both dimensions [33]. Nevertheless, significant disadvantages of this approach include the lengthy analysis time, the challenge of automating the procedure, and the potential for contamination and losses during fraction collection. Online applications use a modulator, usually a valve, to link two dimensions so that fractions eluted from the 1D are automatically collected and sent to the 2D in real time [34]. Numerous ways have been devised to counteract these disadvantages, making online 2D-LC one of the most adaptable methods. Stop-and-go 2D-LC is a methodology that falls in between the offline and online approaches [35,36]. In this system, the 1D flow rate is not restarted until the 2D analysis is completed, and vice versa—the flow in the 2D column is halted when the 1D is operating. The collected fraction is moved from the 1D column to the 2D column during this time. This method has the benefit of allowing for the use of both slow separations in the 2D and large flows in the 1D [5,37] (Figure 2).
Comprehensive heart-cutting 2D-LC
It is possible to resolve target molecules that co-elute in the 1D by collecting and reintroducing a single fraction into the 2D for analysis in the LC-LC mode. By using longer 2D columns and 2D gradients with longer run times than 1D sample times, increased separation efficiency may be achieved since 2D analysis time is independent of 1D run time. Because the remaining peaks from the 1D effluent cannot be examined while the gradient is still flowing in the 2D, this technique’s primary drawback is the loss of information for those peaks that are not analyzed [38]. Hybrid implementations have been created to solve the shortcomings of the previously mentioned methods. One of these is known as multiple heart-cutting two-dimensional liquid chromatography, or MHC for short. It is a great substitute for LC–LC because it can collect and store different fractions of the one-dimensional effluent at the same time as another is being analyzed in the two-dimensional format. The various 1D heart-cutting fractions are kept in several loops linked by extra valves. In this sense, mLC-LC necessitates a sophisticated valve system, with one valve serving as the connection between the 1D and the 2D, and two other valves holding the loops [39]. A unique algorithm based on many criteria drives the mLC-LC procedure used for sample loop filling and the analysis of the stored fractions that follow. The system’s algorithm’s quick commencement of the 2D analysis is an important detail. This implies that any 1D fraction kept in the loops will be immediately moved to the 2D for analysis whenever the 2D is free [40]. A recent study was done, which includes the simultaneous determination of favipiravir and its metabolites with the help of heart-cutting 2D-LC. Herein, they applied this approach to human serum, plasma, and peripheral blood mononuclear cells. Their findings highlighted the reliability of heart-cutting 2D-LC as an efficient approach for therapeutic drug monitoring [41].
Modulation technologies
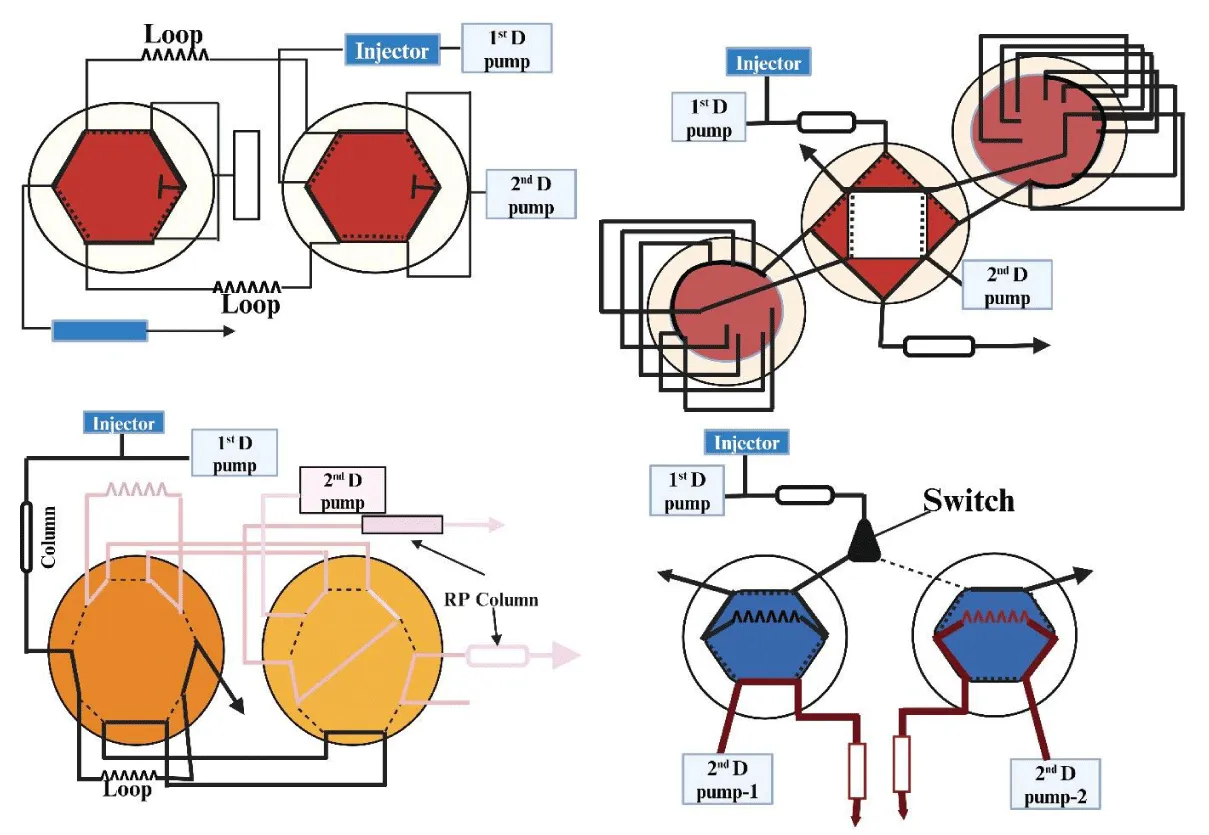
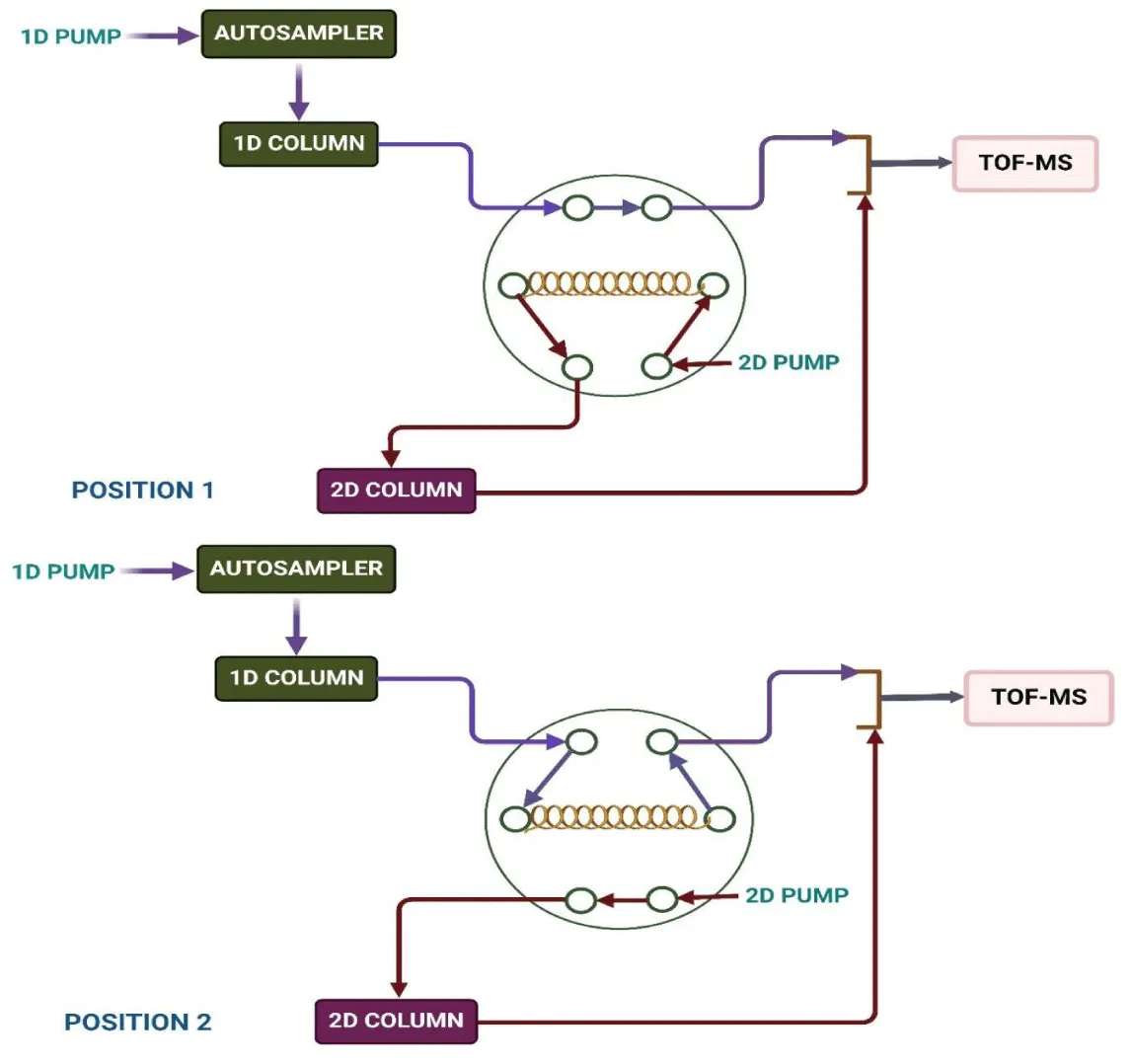
For any 2D-LC technique, the modulator will function as an injector in the 2D, hence choosing the interface used to link 1D and 2D needs to be carefully studied. To prevent losing the separation obtained owing to a potential remixing of the collected fraction, it must be able to effectively collect and transport the 1D effluent. Out of all the available alternatives, electronically controlled switching valves that can automatically collect and transfer fractions between 1D and 2D make up the most commonly used interfaces. These usually involve two-position switching valves with 6, 8, 10, or 12 ports [4,42]. Given the variety of 2D-LC combinations, it is important to carefully consider the selection of the appropriate modulation technique. Many alternative modulation techniques have been devised to date to build a 2D-LC system based on various analytical scenarios. Electric switching valves are the most often used option among the described modulators because they are ideal for realizing automated cutting, collecting, and transferring in 2D-LC. Depending upon the mechanism employed for the modulator, the modulation techniques can be further divided into two categories: i) simple valve-based modulation and ii) valve-linked modulation in combination with assistant technology.
Simple valve-based modulation
In simple valve-based modulation, sample loops frequently directly inject 1D fraction into the 2D column. As a result, the construction of the modulator is quite straightforward. A two-position electronic valve system is used to transition the wastewater from a one-dimensional column to a two-dimensional column once it has been collected by the sample loop. Lynen et al used a temperature gradient for the 1D analysis in the RPLC x RPLC system, where they used pure water as the mobile phase [43].
The combination of IEX and RPLC can also employ simple modulation; however, in these scenarios, the transfer volume is severely constrained to prevent salt precipitation during the 2D separation [44-46]. The mismatch in the mobile phases makes it difficult to accomplish the focusing effect of the transferred analytes on the head of the 2D column in the event of incompatible couplings. However, by lowering the transferring fraction volume, the impact of the mobile phase incompatibility might be mitigated. Additionally, basic modulation could be used in 2D-LC systems with immiscible mobile phases, such as NPLC/RPLC [47,48]. By joining one or more valves, sample loops, steel tubes, and other components, the basic modulation is accomplished. The performance of the separation setup requires a quick separation in 2D; however, this challenge might be somewhat overcome with the application of a stop-flow modulation by stopping the 1D flow for the whole separation of the transferred fraction on the 2D column [5, 49, 50].
Valve modulation with assistant technology
Since simple modulators have a limited effective transferring volume and a large 2D flow, they significantly impair the sensitivity of detection in the 2D in the majority of non-compatible couplings. Moreover, the mismatch incompatibility that causes detrimental consequences in the 2D separation is sometimes unavoidable, even with minimal modulation. Nevertheless, if the 1D wastewater is pre-treated before collection or transfer to the 2D, the problem of a straightforward modulation may be mitigated [51]. The fraction’s pre-treatment can increase the two dimensions’ compatibility, which will help the analytes concentrate more effectively on the 2D column’s head. To date, various pre-treatment methods, including desalting [52], dilution [53,54], evaporation by vacuum [55], dilution, and splitting [53,56] have been utilized in 2D-LC systems.
Single valve modulation with assistant technology
The single-based valve modulation’s pre-treatment procedure is comparatively easy. Most of the time, adding a third pump to provide a make-up flow allows the same setup that was used for the straightforward valve-based modulation to be employed. In cases when the mobile phases (RPLC/RPLC and HILIC/RPLC) are only partially compatible (RPLC/RPLC and HILIC/RPLC), this modulation technique can be used in conjunction with two separation modes. A make-up flow, whose composition is identical to the beginning gradient conditions of the 2D analysis, is added to the 1D effluent to create a dilution of it. Because of this, the transferred fraction is dissolved in an appropriate eluent before being injected into the two-dimensional column [57].
Tian et al developed an orthogonal 2D-LC system by using a vacuum-evaporation loop-type valve interface. First, a bonded CN phase column was utilized in normal-phase liquid chromatography (NPLC), and second, a C18 column was utilized in reversed-phase liquid chromatography (RPLC). The analytes remained on the inner wall of the interface loop after all of the solvents in it evaporated at 90 °C in a vacuum. The second dimension’s mobile phase allowed for the online solvent exchange of a chosen fraction from the first dimension to the second dimension by dissolving the analytes in the loop and injecting them into the secondary column [58].
Multi-valve modulation with assistant technology
Sometimes, more complicated setups are required for incompatible solvent changes and effluent removal in the combination of NPLC/RPLC or IEX/RPLC when there is poor compatibility between mobile phases in two dimensions [59]. Li et al used multi-valve modulation technology when they combined NPLC x RPLC for the analysis of toad skin with a newly developed thermal evaporation-assisted adsorption (TEAA) interface. The solvent incompatibility issue between NPLC and RPLC was effectively resolved by this innovative TEAA interface, which heated the solvent above the solvent boiling point and allowed for the quick removal of organic NPLC solvent [60]. Various types of multi-valve modulation systems are represented in Figure 3.
While the fundamental principles and configurations of two-dimensional liquid chromatography provide the theoretical foundation for enhanced chromatographic separation, the true value of this technique becomes evident in its practical applications. The ability of 2D-LC systems to combine orthogonal separation mechanisms significantly increases peak capacity and analytical resolution, making them particularly suitable for analyzing complex matrices. In recent years, these capabilities have enabled the successful application of 2D-LC techniques in diverse fields, particularly food safety assessment and environmental monitoring. The following sections discuss representative applications of 2D-LC in these areas, highlighting how different chromatographic configurations have been employed to address specific analytical challenges.
2D-LC in Food Analysis
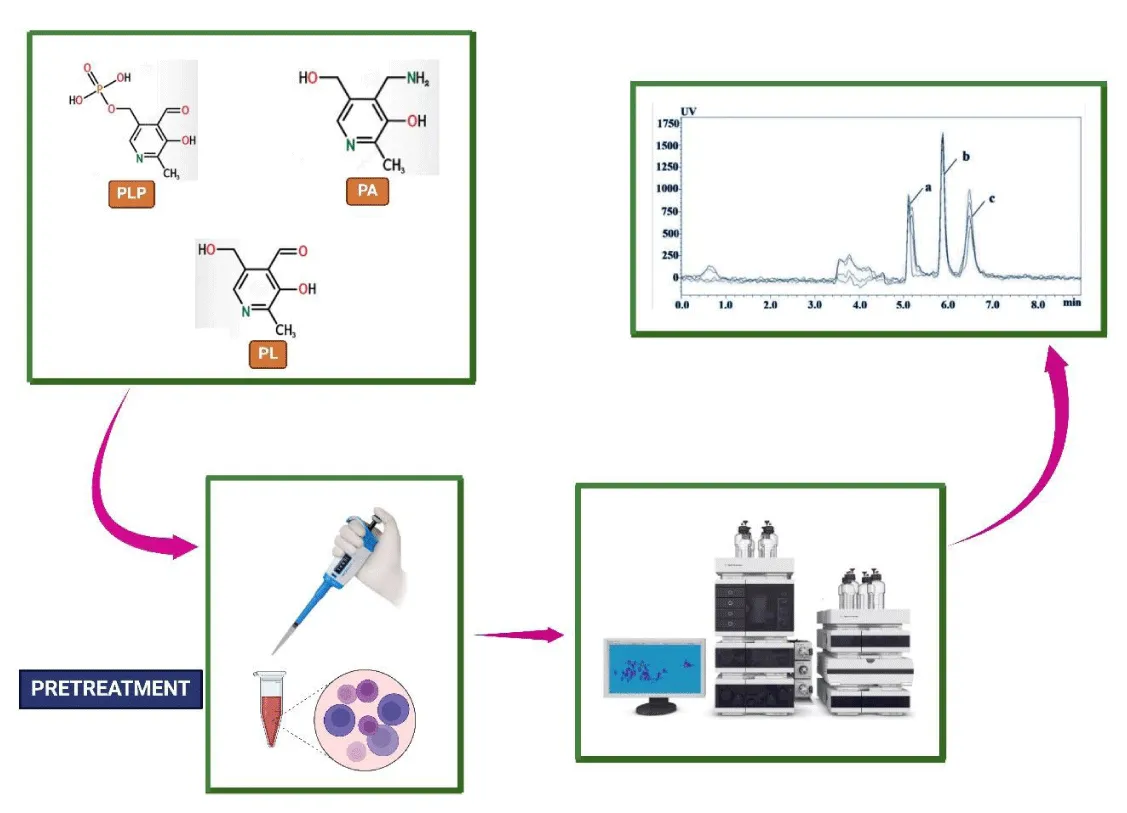
Pyridoxal 5’-phosphate, 4-pyridoxine acid, and pyridoxal
Yang et al used a novel 2D-LC/UV method for quantitative detection of Pyridoxal 5’- phosphate, 4-Pyridoxine acid, and Pyridoxal inside the animal plasma. They stated that variations in the content and level of Vit B6 can alter the complete physiological condition of the body. They used a two-dimensional liquid chromatography-UV detector (2D-LC-UV) to establish a method for the simultaneous detection of PLP, PA, and PL for the first time. Before derivatization, PLP, PA, and PL were extracted using plasma using the following ratio: 0.6 M TCA: ultrapure water = 1:2:3 (v/v/v). PLP, PA, and PL had respective detection limits of 0.1, 0.2, and 4 nmol/L. The system has a nice peak shape, large loading capacity, and great resolution, according to the results [61]. The method by which PLP, PA, and PL can be detected is listed in Figure 4.
Comparative evaluation of 2D-LC methods in food analysis
Two-dimensional liquid chromatography (2D-LC) techniques have been used in various food types, including beverages, grains, bio-oils, and dairy products. Many studies show that the effectiveness of 2D-LC in food analysis can differ based on the nature of the analytes and the complexity of the matrix [1,6,10,20]. A comparison of the methods found in the literature reveals several important trends.
For example, 2D-LC systems combined with high-resolution mass spectrometry (2D-LC-HRMS) usually offer better sensitivity and selectivity for detecting trace contaminants. This is especially true for mycotoxins and pesticide residues in food samples [20,64,66]. Research using LC×LC-HRMS has shown detection limits in the microgram per kilogram range, which is adequate for regulatory monitoring of contaminants in food products [64,67]. In contrast, 2D-LC methods combined with ultraviolet detection (2D-LC-UV) work better for identifying more common analytes like vitamins and metabolic compounds, where very low detection limits are not necessary [28,61]
Likewise, comprehensive two-dimensional liquid chromatography (LC×LC) provides a greater peak capacity and better separation efficiency compared to heart-cutting 2D-LC methods [18,19,39]. LC×LC is particularly useful for analyzing complex mixtures, like phenolic compounds in beverages or peptide profiles in protein hydrolysates. However, heart-cutting methods are often preferred for focused analysis of specific analytes because they allow for longer second-dimension separations and improved sensitivity for selected fractions [9,38].
Overall, the choice of 2D-LC setup depends on the analytical goal. Comprehensive LC×LC methods are more suitable for untargeted screening and profiling of complex mixtures, while heart-cutting techniques are better for targeted measurement of specific compounds [9,18,19]. These findings emphasize the need to choose the right 2D-LC configuration based on the analytical needs of the food matrix being studied [1,10].
Asparagine and aspartic acid
Goyon et al utilized an automated and robust multi-dimensional liquid chromatography and mass spectrometry for the changing characterization of various therapeutic antibodies. Specifically, the characterization of charge variations separated by Cation Exchange Chromatography (CEX) is a laborious procedure that is often carried out by peptide mapping after an offline manual fraction collection. A general online multi-dimensional LC/MS methodology was created for the characterization of several monoclonal antibody (mAb) isotypes and a bi-specific antibody (BsAb) to increase the effectiveness of this laborious method. Fractions collected from 1D CEX analysis were consecutively reduced on a 2D reversed-phase liquid chromatography (RPLC) column (polyphenyl). Comparisons were made between 3D trypsin cartridges and 2D RPLC columns from various vendors, as well as the results of reducing agents. Additionally, the effects of 2D and 4D RPLC column temperatures as well as 2D RPLC column mass load were methodically investigated. The multi-dimensional LC/MS system shown in this work is a reliable instrument for the online digestion of proteins and exhibits great reproducibility under ideal circumstances. This online methodology appears to have fewer errors than the manual offline procedure, as seen by the lower levels of deamidation and isomerization observed at certain aspartic acid and asparagine residues using the online approach. With the use of the multi-dimensional LC/MS method shown here, therapeutic antibodies may be quickly and automatically characterized online without the requirement for sample pre-treatment or offline fraction collection [62].
Bio-oils obtained from different biomasses
Lazzari et al performed a quantitative analysis of hydrophilic phases of bio-oils derived from heating of various biomasses by using 2D-LC. Herein, they stated that under the circular economy concept, agrifood by-products are ideal c OJABC-26-RW-148-Q2andidates for further processing to increase their value. Even though food waste is produced in large quantities all around the world, it may nevertheless contain useful compounds. The use of quantitative online comprehensive 2D-LC (LC × LC) was done to characterize several aqueous phases of various bio-oils. The biomass of peanut shells, rice husks, peach cores, wasted coffee grounds, and eucalyptus sawdust was examined. With LODs and LOQs as low as 0.05 g mL−1 and 0.16 g mL−1, respectively, the developed quantitative LC × LC technique demonstrated extremely excellent linearity, accuracy, repeatability, and recovery [63].
Mycotoxins in Pu-erh tea
Bogdanova et al used 2D-LC-HRMS (High-Resolution Mass Spectrometry) for the simultaneous detection of 70 emerging mycotoxins in Pu-erh tea. They showed that using two-dimensional liquid chromatography combined with high-resolution mass spectrometry, 70 controlled and emergent mycotoxins in Pu-erh tea may be monitored simultaneously, as seen in Figure 5. An occurrence study revealed actual contamination with 19 out of the 70 mycotoxins analyzed; the detected concentrations ranged from 0.5 (for ENN B1) to 8946 μg/kg. The developed method was successfully validated by the performance criteria set in EU guidelines and applied to the analysis of 20 commercially available Pu-erh tea samples [64].
Hydrolysates in casein
Hohme et al developed a method for the characterization of bitter peptides present in casein hydrolysates with the help of 2D-LC. Casein hydrolysates are essential dietary supplements for both sports nutrition and the elderly. However, the use of so-called bitter peptides is restricted because of their enzymatic synthesis. Therefore, they described a unique approach for evaluating the bitterness of hydrolysate samples based on their elution pattern by combining two separation techniques utilizing comprehensive two-dimensional liquid chromatography. A thorough assessment of the sample’s peptide size and relative hydrophobicity was possible by using a size exclusion column in the first dimension and a reversed-phase column in the second. The sensory evaluation and the outcomes for various casein hydrolysates were connected. It was discovered that hydrolysates that exhibit a greater degree of bitterness also include a greater quantity of peptides with a molecular weight of less than 6.5 kDa and strong hydrophobicity [65].
Vitamin A, D, and E
Zhang et al studied and implemented a method for fast and rapid determination of Vitamin A, D, and E homologues in food samples by a solid phase extraction technique combined with 2D-LC. Through saponification, the target analytes in this approach were freed from their lipoprotein-coated form. The saponified solution was then immediately injected for enrichment, purification, separation, and quantification into the system that was built by combining online SPE with heart-cutting 2D-LC. The online SPE technique was enhanced using online dilution to reduce the impact of the solvent on the first-column separation. The first column in the study of the four E homologues and vita A was PFP. PAH was utilized as the second column in the analysis of vitamin D. The analytes’ strong linearity (R2>0.9999) and good reproducibility (RSD < 2%, n = 5) were demonstrated by the findings. As determined by S/N = 10:1, the limits of quantification for vit A, D2, D3, and α-, β-, γ-, and δ-vitamin E were 0.030, 0.013, 0.014, 0.51, 0.33, 0.31, and 0.33 ng, in that order. The range of spiked recoveries was 93.29% to 103.66% [28].
Pesticides in corn-based products
Arena et al used a technique known as solid-liquid extraction followed by 2D-LC/MS for the detection of pesticide residues in five different corn-based products. Thus, their research offered a unique analytical technique for the simultaneous detection of one hundred and thirteen multiclass pesticides in maize products using comprehensive 2D-LC/MS. The limit of quantification was between 0.3 and 91.6 μg/L, whereas the limit of detection varied between 0.2 and 30.2 μg/L, all lying within the required limits. Precision, as determined by relative standard deviation, was less than 15.1%, whereas accuracy varied from 60% to 120%. Pesticides in five sample maize products (tortilla, corn flakes, corn cake, starch, and polenta) were analyzed to show the effectiveness of the procedure. At last, sixteen products were detected, and all of them were within the residual limits [66].
112 pesticides in corn-based products
Pozo et al developed and validated a comprehensive 2D-LC/MS for the detection of pesticides in corn-related products. For the first time, a multi-class study of 112 pesticides in corn-based products using tandem mass spectrometry and extensive 2D-LC was offered. The intra-day and inter-day precision were less than 12.9% and 15.1%, respectively, and the limits of quantification values were lower than those set by European legislation. More than 70% of the analytes had recoveries with standard deviation values less than 20% and recoveries in the range of 70% to 120%. Furthermore, the values of the matrix impact ranged from 13% to 161%. Three pesticides were found in trace amounts in both of the samples when the same procedure was used to analyze actual samples [67].
Wine phenolics
Wicht et al utilized 2D-LC x RPLC for the analysis of wine phenolics. Here, they stated that often highly complex, phenolic extracts from natural products contain compounds spanning a wide range of molecular properties, making phenolic compounds an intriguing class of natural products due to their potential contribution to the health benefits of foods and beverages, as well as their use as a bio-source of organic (aromatic) building blocks. Phenolics have been the subject of several 1D-LC and mass spectrometric techniques; nonetheless, their complexity invariably makes identification and purification difficult. Through online comprehensive 2D-LC linked to a photodiode array and mass spectrometric detector, new insights into the composition of phenolic extracts may be acquired [68]. More applications of 2D-LC related to food analysis are mentioned in Table 1.
2D-LC in Environmental Analysis
Elucidation of degradation caused by carbamazepine
Bandini et al developed a temperature-responsive comprehensive 2D-LC coupled with HRMS for the representation of oxidatively degraded products that were formed from the oxidation of organic pollutants like carbamazepine (CBZ). To better understand the degradation cascade and enable real-time monitoring of the appearance and disappearance of various degradation products, the TRLC×RPLC combination effectively separates and identifies CBZ and its degradation products while offering improved selectivity over the individual TRLC or RPLC separations. Their research focused on the oxidative degradation of CBZ, a compound that is of environmental concern due to its persistence in water bodies. This article demonstrates the compatibility with high-resolution mass spectrometry, which allows the identification of 21 products related to CBZ, nine of which were not previously reported in studies on CBZ degradation [73].
Degradation of pharmaceuticals in water
Souza et al validated a 2D-LC×HRMS method to detect photoelectrochemical degradation of various pharmaceuticals and their degradation products in water samples. Given this, advanced oxidation processes (AOPs) have become a viable option for treating quaternary wastewater because they effectively break down resistant components by producing very reactive free radicals. However, chemical characterization of potentially toxic leftovers is necessary to prevent dangerous materials from contaminating natural water bodies. Several medications were tested in ultrapure water and wastewater simulation, and after 180 minutes of treatment, good removal efficiencies (total >95%) were attained. After a total analysis time of 50 min, the LC×HRMS allowed the detection of a total of 12 venlafaxines, 7 trimethoprim, and 10 ciprofloxacin analogues [74].
Organic atmospheric tracers in ice cores
Beschnitt et al used a comprehensive heart-cutting 2D-LC approach for analyzing different organic atmospheric traces inside ice ores. The results of a non-target screening were restricted to signals of chemicals within a certain polarity range based on the stationary phases of the columns that were used when mass spectrometry and chromatography were coupled. A second column with distinct functionality was incorporated in comprehensive two-dimensional liquid chromatography (LC x LC) to allow for the examination of a wider variety of analytes. When the method was used for snow samples gathered at the high-alpine research station Jungfraujoch, it produced a total of 270 signals. Method validation demonstrated sufficient repeatability and detection limits for two chosen void volume indicators. 175 signals were only found in the two-dimensional technique, of which 23 were found in the second dimension, as compared to the one-dimensional HILIC methodology. The findings demonstrated that one-dimensional chromatography was insufficient to cover the whole spectrum of substances, and the two-dimensional technique that was devised will shorten the analysis time while improving the information content from non-target screening [75].
Antidepressants in wastewater
Zhong et al developed a method for the simultaneous identification of various antidepressants and also their enantiomers inside wastewater by using heart-cutting 2D-LC coupled with solid-phase extraction (SPE). Target analytes were able to convert across 2D-LCs well, with a transfer rate ranging from 90.57% to 98.58%. Three antidepressants in wastewater were significantly preconcentrated under ideal circumstances using freeze-drying and SPE, which enhanced the efficiency of the procedure. Bupropion, fluoxetine, and their racemates and enantiomers all showed good linearity in the 0.10–30.00 ng/mL (R2≥0.9986) range, and their LODs and LOQs were 0.0183–0.0549 ng/mL and 0.0661–0.1831 ng/mL, respectively. Therefore, the suggested method offers an effective way to track chiral pharmaceuticals and their enantiomers in wastewater, making it easier to analyze the pollution caused by chiral drugs in the environment and conduct a regional survey on drug misuse [76].
Phosphorus compounds in soil
Gachumi et al, with the help of 2D-LC-MS/MS, detected and analyzed phosphorus (Po) compounds in soil, as seen in Figure 6. As a result, a technique known as 2D-IC-MS/MS was developed to determine the Po concentrations in soil. Before the second dimension’s chromatographic separation and detection, the first dimension preconcentrated P compounds in a trap column and diverted early eluting anions to trash. The P substances under study, which were primarily nucleotides, had a linear range of 0.05 to 50 ng/mL. In calcareous subsoil samples, the majority of P compounds were found and measured in concentration ranges of 0.70 – 51.78 ng/g. The developed method produced chromatographic separation, which made it possible to identify isobars/isomers and isotopologues that unambiguously interfere with MS detection [77].
Dichloronitrophenol in industrial wastewater
Purschke et al, in the current investigation, identified various unknown compounds in industrial wastewater by using 2D chromatography and non-target screening. The identification of compounds of interest is highly time-intensive and sometimes ineffective due to the great diversity of the matrix. To get around this restriction, a prioritization technique was created to determine which chemicals in industrial wastewater should be prioritized as “known unknowns,” or substances that are regularly found but not characterized. The technique uses high-resolution mass spectrometry (HRMS) for the second dimension and ultraviolet (UV) detection for the first dimension in an offline two-dimensional 2D-LC approach. An identifying procedure for one “known unknown” is provided as a proof of concept. By using the UV spectrum, mass fragmentation, retention time in two dimensions, precise mass, and 1H-NMR, the chemical was determined to be a dichlorodinitrophenol isomer [78].
Aromatic hydrocarbons in water samples
Armutcu et al, with the use of reversed-phase liquid chromatography (RPLC) and monolithic columns, invented an online heart-cutting two-dimensional liquid chromatography (2D-LC) technique. An affinity-based monolithic column (20 cm × 4.6 mm I.D.) was used to complete the first dimension of 2D-HPLC, and a Pinnacle II PAH column (50 mm × 4.6 mm I.D.) was used for the second dimension. When every factor is taken into account, the data show that the 2D-HPLC system, which was developed by combining the P(HEMA-MAPA) and Pinnacle II PAH columns, is trustworthy, stable, and highly capable of extracting polycyclic aromatic hydrocarbons from water samples [79]. Various other applications of 2D-LC in environmental analysis are mentioned in Table 2.
Comparative perspective on environmental applications of 2D-LC
Recent uses of two-dimensional liquid chromatography in environmental analysis show its clear benefits in detecting low-level pollutants and complex environmental contaminants [14,15].. Environmental samples like wastewater, soil extracts, and atmospheric deposits contain a variety of organic compounds with different chemical properties. This variety makes detailed analysis difficult with traditional one-dimensional chromatography [14].
Combining 2D-LC with high-resolution mass spectrometry has been especially effective for identifying pharmaceuticals, micropollutants, and breakdown products in environmental samples [80,81]. For instance, research on pharmaceutical contaminants in wastewater indicates that LC×LC-HRMS greatly enhances detection compared to conventional LC-MS methods [80,81]. This improvement allows for identifying multiple transformation products that occur during water treatment [73,74].
Additionally, heart-cutting 2D-LC techniques have effectively analyzed chiral pharmaceuticals and their metabolites in wastewater samples [76]. These focused methods allow for the selective extraction of analytes from complex mixtures before further chromatographic separation [38,76]. This process improves sensitivity and decreases interference from the matrix.
Another important application is the identification of unknown compounds in industrial wastewater using offline 2D-LC paired with non-target screening methods [78]. This strategy allows for prioritizing and identifying the structure of previously unknown environmental contaminants.
In summary, the studies reviewed show that 2D-LC provides key advantages for environmental analysis, especially when identifying complex mixtures or low-level contaminants [14,15,81]. The increased peak capacity, better orthogonality, and greater compatibility with modern detection systems make 2D-LC a vital tool for current environmental monitoring [18,82].
Conclusion
Two-dimensional liquid chromatography (2D-LC) has revolutionized the field of food analysis by addressing the limitations of one-dimensional liquid chromatography (1D-LC), particularly in the separation and identification of complex food matrices. This advanced technique significantly enhances the resolution of closely related and co-eluting species, which are often challenging to separate using traditional methods. Coupling 2D-LC with mass spectrometry (MS) further amplifies its analytical capabilities, offering increased sensitivity, specificity, and lower detection limits, which are crucial for the accurate structural identification of food components. The application of 2D-LC-MS extends beyond food analysis into environmental monitoring, where it plays a critical role in detecting and characterizing organic micropollutants (OMPs) at trace levels, impurities in therapeutic oligonucleotides, and various pharmaceuticals in wastewater. This ability to comprehensively analyze complex environmental and food samples ensures that food safety, quality, and regulatory standards are met, ultimately protecting public health. As the demand for high-resolution analytical techniques continues to grow, 2D-LC, particularly when integrated with MS, is expected to become an indispensable tool in both food and environmental sciences. Future advancements in 2D-LC technology will likely further enhance its applicability, making it a cornerstone of analytical practices in these fields.
Conflict of interest
The author declares that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper hence, the author finds no conflict of interest.
Data availability statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
- Liang L, Duan W, Zhao C, Yuyu Z. Recent development of two-dimensional liquid chromatography in food analysis. Food Analytical Methods. 2022;1-12. Available from: https://doi.org/10.1007/s12161-021-02190-2
- Delgado AM, Issaoui M, Chammem N. Analysis of main and healthy phenolic compounds in foods. Journal of AOAC International. 2019;102(5):1356-1364. Available from: https://doi.org/10.5740/jaoacint.19-0128
- Cavaliere C, Capriotti AL, La Barbera G, Montone CM, Piovesana S, Laganà A. Liquid chromatographic strategies for separation of bioactive compounds in food matrices. Molecules. 2018;23(12):3091. Available from: https://www.mdpi.com/1420-3049/23/12/3091
- Cacciola F, Arena K, Mandolfino F, Donnarumma D, Dugo P, Mondello L. Reversed-phase versus hydrophilic interaction liquid chromatography as the first dimension of comprehensive two-dimensional liquid chromatography systems for the elucidation of the polyphenolic content of food and natural products. Journal of Chromatography A. 2021;1645:462129. Available from: https://doi.org/10.1016/j.chroma.2021.462129
- Dugo P, Fawzy N, Cichello F, Cacciola F, Donato P, Mondello L. Stop-flow comprehensive two-dimensional liquid chromatography combined with mass spectrometric detection for phospholipid analysis. Journal of Chromatography A. 2013;1278:46-53. Available from: https://doi.org/10.1016/j.chroma.2012.12.042
- Cacciola F, Donato P, Sciarrone D, Dugo P, Mondello L. Comprehensive liquid chromatography and other liquid-based comprehensive techniques coupled to mass spectrometry in food analysis. Analytical Chemistry. 2017;89(1):414-429. Available from: https://pubs.acs.org/doi/abs/10.1021/acs.analchem.6b04370
- Valdés A, Álvarez-Rivera G, Socas-Rodríguez B, Herrero M, Ibáñez E, Cifuentes A. Foodomics: Analytical opportunities and challenges. Analytical Chemistry. 2021;94(1):366-381. Available from: https://pubs.acs.org/doi/10.1021/acs.analchem.1c04678
- Alam MS, McGregor LA, Harrison RM. A review of organic aerosol speciation by comprehensive two-dimensional gas chromatography. TrAC Trends in Analytical Chemistry. 2024:117718. Available from: https://research.birmingham.ac.uk/en/publications/a-review-of-organic-aerosol-speciation-by-comprehensive-two-dimen/
- Caño-Carrillo I, Gilbert-López B, Montero L, Martínez-Piernas AB, García-Reyes JF, et al. Comprehensive and heart-cutting multidimensional liquid chromatography-mass spectrometry and its applications in food analysis. Mass Spectrometry Reviews. 2023. Available from: https://doi.org/10.1002/mas.21845
- Montero L, Herrero M. Two-dimensional liquid chromatography approaches in Foodomics–a review. Analytica Chimica Acta. 2019;1083:1-18. Available from: https://doi.org/10.1016/j.aca.2019.07.036
- Di Stefano V, Avellone G, Bongiorno D, Cunsolo V, Muccilli V, Sforza S, et al. Applications of liquid chromatography-mass spectrometry for food analysis. Journal of Chromatography A. 2012;1259:74-85. Available from: https://doi.org/10.1016/j.chroma.2012.04.023
- Lopes D, Morés L, Silva M, Schneider M, Merib J, Carasek E. Determination of hormones in urine by hollow fiber microporous membrane liquid-liquid extraction associated with 96-well plate system and HPLC-FLD detection. Journal of Chromatography B. 2022;1207:123406. Available from: https://doi.org/10.1016/j.jchromb.2022.123406
- Li W, Huang J, Zheng L, Liu W, Fan L, Sun B. A fast stop-flow two-dimensional liquid chromatography-tandem mass spectrometry and its application in food-derived protein hydrolysates. Food Chemistry. 2023;406:135000. Available from: https://doi.org/10.1016/j.foodchem.2022.135000
- Duarte RM, Brandão PF, Duarte AC. Multidimensional chromatography in environmental analysis: Comprehensive two-dimensional liquid versus gas chromatography. Journal of Chromatography A. 2023:464288. Available from: https://doi.org/10.1016/j.chroma.2023.464288
- Pérez-Fernández V, Mainero Rocca L, Tomai P, Fanali S, Gentili A. Recent advancements and future trends in environmental analysis: Sample preparation, liquid chromatography and mass spectrometry. Analytica Chimica Acta. 2017;983:9-41. Available from: https://doi.org/10.1016/j.aca.2017.06.029
- Zhang T, Liang X, Wang L, Wang S, Liu X, Yong Y, et al. An ionic liquid assisted hydrogel functionalized silica stationary phase for mixed-mode liquid chromatography. Chinese Chemical Letters. 2024:109889. Available from: https://doi.org/10.1016/j.cclet.2024.109889
- Shah PA, Shrivastav PS, Sharma V. Multidimensional chromatography platforms: status and prospects. Bioanalysis. 2021;13(14):1083-1086. Available from: https://doi.org/10.4155/bio-2021-0118
- Pirok BWJ, Stoll DR, Schoenmakers PJ. Recent developments in two-dimensional liquid chromatography: fundamental improvements for practical applications. Analytical Chemistry. 2018;91(1):240-263. Available from: https://doi.org/10.1021/acs.analchem.8b04841
- Stoll DR, Carr PW. Two-dimensional liquid chromatography: a state of the art tutorial. Analytical Chemistry. 2017;89(1):519-531. Available from: https://pubs.acs.org/doi/10.1021/acs.analchem.6b03506
- Arena K, Mandolfino F, Cacciola F, Dugo P, Mondello L. Multidimensional liquid chromatography approaches for analysis of food contaminants. Journal of Separation Science. 2021;44(1):17-34. Available from: https://doi.org/10.1002/jssc.202000754
- Tranchida PQ, Donato P, Cacciola F, Beccaria M, Dugo P, et al. Potential of comprehensive chromatography in food analysis. TrAC Trends in Analytical Chemistry. 2013;52:186-205. Available from: https://doi.org/10.1016/j.trac.2013.07.008
- Dong X, Yang J, Wang QY, Zhen XT, Liu FM, Zheng H, et al. Microextraction assisted multiple heart-cutting and comprehensive two-dimensional liquid chromatography hyphenated to Q-TOF/MS for the determination of multiclass compounds from Dendrobium species. Microchemical Journal. 2020;157:105097. Available from: https://doi.org/10.1016/j.microc.2020.105097
- Nardiello D, Melfi MT, Pignatelli C, Centonze D. Enhancing online protein isolation as intact species from soy flour samples by actively modulated two-dimensional liquid chromatography (2D-LC). Journal of Pharmaceutical and Biomedical Analysis. 2020;179:112976. Available from: https://doi.org/10.1016/j.jpba.2019.112976
- Zhang Y, Chen M, Li L, Lv Y, Ma Q. Recent advances in microextraction techniques using sustainable green solvents for mass spectrometry analysis. TrAC Trends in Analytical Chemistry. 2023:117412. Available from: https://doi.org/10.1016/j.trac.2023.117412
- Campone L, Rizzo S, Piccinelli AL, Celano R, Pagano I, Russo M, et al. Determination of mycotoxins in beer by multi-heart-cutting two-dimensional liquid chromatography-tandem mass spectrometry method. Food Chemistry. 2020;318:126496. Available from: https://doi.org/10.1016/j.foodchem.2020.126496
- Russo M, Cacciola F, Arena K, Mangraviti D, de Gara L, Dugo P, et al. Characterization of the polyphenolic fraction of pomegranate samples by comprehensive two-dimensional liquid chromatography coupled to mass spectrometry detection. Natural Product Research. 2020;34(1):39-45. Available from: https://doi.org/10.1080/14786419.2018.1561690
- Muller M, Tredoux AG, de Villiers A. Application of kinetically optimized online HILIC×RP-LC methods hyphenated to high-resolution MS for the analysis of natural phenolics. Chromatographia. 2019;82:181-196. Available from: https://doi.org/10.1007/s10337-018-3662-6
- Zhang Y, Lin Y, Yang X, Chen G. Fast determination of vitamin A, vitamin D, and vitamin E in food by online SPE combined with heart-cutting two-dimensional liquid chromatography. Journal of Food Composition and Analysis. 2021;101:103983. Available from: https://doi.org/10.1016/j.jfca.2021.103983
- Zhang Y, Li X, Wang S, Liu X, Chen M, et al. Enabling the ion-mobility separation and the alternate data-dependent and data-independent acquisitions (HDDIDDA): Its combination with off-line two-dimensional liquid chromatography for comprehensively characterizing the multicomponents from Compound Danshen Dripping Pill. Analytica Chimica Acta. 2022;1193:339320. Available from: https://doi.org/10.1016/j.aca.2021.339320
- Fuente-Ballesteros A, Bernal J, Ares AM, Valverde S. Development and validation of a green analytical method for simultaneously determining plasticizers residues in honey from different botanical origins. Food Chemistry. 2024:139888. Available from: https://doi.org/10.1016/j.foodchem.2024.139888
- Berton P, Lana NB, Ríos JM, García-Reyes JF, Altamirano JC, et al. State of the art of environmentally friendly sample preparation approaches for determination of PBDEs and metabolites in environmental and biological samples: A critical review. Analytica Chimica Acta. 2016;905:24-41. Available from: https://doi.org/10.1016/j.aca.2015.11.009
- Cacciola F, Farnetti S, Dugo P, Marriott PJ, Mondello L. Comprehensive two-dimensional liquid chromatography for polyphenol analysis in foodstuffs. Journal of Separation Science. 2017;40(1):7-24. Available from: https://doi.org/10.1002/jssc.201600704
- Horváth K, Fairchild J, Guiochon G. Optimization strategies for off-line two-dimensional liquid chromatography. Journal of Chromatography A. 2009;1216(12):2511-2518. Available from: https://doi.org/10.1016/j.chroma.2009.01.064
- Česla P, Křenková J. Fraction transfer process in online comprehensive two-dimensional liquid-phase separations. Journal of Separation Science. 2017;40(1):109-123. Available from: https://doi.org/10.1002/jssc.201600921
- Chapel S, Heinisch S. Strategies to circumvent the solvent strength mismatch problem in online comprehensive two-dimensional liquid chromatography. Journal of Separation Science. 2022;45(1):7-26. Available from: https://doi.org/10.1002/jssc.202100534
- Groeneveld G, Dunkle MN, Pursch M, Mes EPC, Schoenmakers PJ, et al. Investigation of the effects of solvent-mismatch and immiscibility in normal-phase × aqueous reversed-phase liquid chromatography. Journal of Chromatography A. 2022;1665:462818. Available from: https://doi.org/10.1016/j.chroma.2022.462818
- Gutiérrez-Escobar R, Aliaño-González MJ, Cantos-Villar E. Wine polyphenol content and its influence on wine quality and properties: A review. Molecules. 2021;26(3):718. Available from: https://doi.org/10.3390/molecules26030718
- Pursch M, Buckenmaier S. Loop-based multiple heart-cutting two-dimensional liquid chromatography for target analysis in complex matrices. Analytical Chemistry. 2015;87(10):5310-5317. Available from: https://doi.org/10.1021/acs.analchem.5b00492
- Pardon M, Reis R, de Witte P, Chapel S, Cabooter D. Detailed comparison of in-house developed and commercially available heart-cutting and selective comprehensive two-dimensional liquid chromatography systems. Journal of Chromatography A. 2024;1713:464565. Available from: https://doi.org/10.1016/j.chroma.2023.464565
- Arena K, Cacciola F, Dugo L, Dugo P, Mondello L. Determination of the metabolite content of Brassica juncea cultivars using comprehensive two-dimensional liquid chromatography coupled with a photodiode array and mass spectrometry detection. Molecules. 2020;25(5):1235. Available from: https://www.mdpi.com/1420-3049/25/5/1235
- Sánchez-Hernández A, García-Gómez D, Pérez Pavón JL, Rodríguez-Gonzalo E. Simultaneous determination of favipiravir and surrogates of its metabolites using heart-cutting bidimensional liquid chromatography (2D-LC). Analytical Biochemistry. 2024;684:115375. Available from: https://doi.org/10.1016/j.ab.2023.115375
- Montero L, Ibáñez E, Russo M, Rastrelli L, Cifuentes A, Herrero M. Focusing and non-focusing modulation strategies for the improvement of on-line two-dimensional hydrophilic interaction chromatography × reversed phase profiling of complex food samples. Analytica Chimica Acta. 2017;985:202-212. Available from: https://doi.org/10.1016/j.aca.2017.07.013
- Baert M, Martens S, Desmet G, de Villiers A, Du Prez F, Lynen F. Enhancing the possibilities of comprehensive two-dimensional liquid chromatography through hyphenation of purely aqueous temperature-responsive and reversed-phase liquid chromatography. Analytical Chemistry. 2018;90(8):4961-4967. Available from: https://pubs.acs.org/doi/abs/10.1021/acs.analchem.7b04914
- Vonk RJ, Wouters S, Barcaru A, Vivó-Truyols G, Eeltink S, de Koning LJ, et al. Post-polymerization photografting on methacrylate-based monoliths for separation of intact proteins and protein digests with comprehensive two-dimensional liquid chromatography hyphenated with high-resolution mass spectrometry. Analytical and Bioanalytical Chemistry. 2015;407:3817-3829. Available from: https://doi.org/10.1007/s00216-015-8615-4
- Sandra K, Vanhoenacker G, Vandenheede I, Steenbeke M, Joseph M, Sandra P. Multiple heart-cutting and comprehensive two-dimensional liquid chromatography hyphenated to mass spectrometry for the characterization of the antibody-drug conjugate ado-trastuzumab emtansine. Journal of Chromatography B. 2016;1032:119-130. Available from: https://doi.org/10.1016/j.jchromb.2016.04.040
- Kwiatkowski M, Krösser D, Wurlitzer M, Steffen P, Barcaru A, Krisp C, et al. Application of displacement chromatography to online two-dimensional liquid chromatography coupled to tandem mass spectrometry improves peptide separation efficiency and detectability for the analysis of complex proteomes. Analytical Chemistry. 2018;90(16):9951-9958. Available from: https://doi.org/10.1021/acs.analchem.8b02189
- Cacciola F, Giuffrida D, Mondello L. Application of comprehensive two-dimensional liquid chromatography for carotenoid analysis in red mamey (Pouteria sapote) fruit. Food Analytical Methods. 2016;9:2335-2341. Available from: https://doi.org/10.1007/s12161-016-0416-7
- Dugo P, Herrero M, Kumm T, Giuffrida D, Dugo G, Mondello L. Comprehensive normal-phase × reversed-phase liquid chromatography coupled to photodiode array and mass spectrometry detection for the analysis of free carotenoids and carotenoid esters from mandarin. Journal of Chromatography A. 2008;1189(1-2):196-206. Available from: https://doi.org/10.1016/j.chroma.2007.11.116
- Xu J, Sun-Waterhouse D, Qiu C, Zhao M, Sun B, Lin L, et al. Additional band broadening of peptides in the first size-exclusion chromatographic dimension of an automated stop-flow two-dimensional high-performance liquid chromatography. Journal of Chromatography A. 2017;1521:80-89. Available from: https://agris.fao.org/search/en/providers/122535/records/65e01c67b766d82b180412a4
- Xu J, Zheng L, Lin L, Sun B, Su G, Zhao M. Stop-flow reversed-phase liquid chromatography × size-exclusion chromatography for separation of peptides. Analytica Chimica Acta. 2018;1018:119-126. Available from: https://doi.org/10.1016/j.aca.2018.02.025
- Chen Y, Montero L, Schmitz OJ. Advance in on-line two-dimensional liquid chromatography modulation technology. TrAC Trends in Analytical Chemistry. 2019;120:115647. Available from: https://doi.org/10.1016/j.trac.2019.115647
- Pirok BWJ, den Uijl MJ, Moro G, Berbers SVJ, Croes CJM, van Bommel MR, et al. Characterization of dye extracts from historical cultural-heritage objects using state-of-the-art comprehensive two-dimensional liquid chromatography and mass spectrometry with active modulation and optimized shifting gradients. Analytical Chemistry. 2019;91(4):3062-3069. Available from: https://doi.org/10.1021/acs.analchem.8b05469
- Stoll DR, Talus ES, Harmes DC, Zhang K. Evaluation of detection sensitivity in comprehensive two-dimensional liquid chromatography separations of an active pharmaceutical ingredient and its degradants. Analytical and Bioanalytical Chemistry. 2015;407:265-277. Available from: https://doi.org/10.1007/s00216-014-8036-9
- Vonk RJ, Gargano AF, Davydova E, Dekker HL, Eeltink S, de Koning LJ, et al. Comprehensive two-dimensional liquid chromatography with stationary-phase-assisted modulation coupled to high-resolution mass spectrometry applied to proteome analysis of Saccharomyces cerevisiae. Analytical Chemistry. 2015;87(10):5387-5394. Available from: https://doi.org/10.1021/acs.analchem.5b00708
- Yang L, Lv P, Ai W, Li L, Shen S, Nie H, et al. Lipidomic analysis of plasma in patients with lacunar infarction using normal-phase/reversed-phase two-dimensional liquid chromatography–quadrupole time-of-flight mass spectrometry. Analytical and Bioanalytical Chemistry. 2017;409:3211-3222. Available from: https://doi.org/10.1007/s00216-017-0261-6
- Stephan S, Hippler J, Köhler T, Deeb AA, Schmidt TC, Schmitz OJ. Contaminant screening of wastewater with HPLC-IM-qTOF-MS and LC+ LC-IM-qTOF-MS using a CCS database. Analytical and Bioanalytical Chemistry. 2016;408:6545-6555. Available from: https://doi.org/10.1007/s00216-016-9820-5
- Creese ME, Creese MJ, Foley JP, Cortes HJ, Hilder EF, Shellie RA, et al. Longitudinal on-column thermal modulation for comprehensive two-dimensional liquid chromatography. Analytical Chemistry. 2017;89(2):1123-1130. Available from: https://doi.org/10.1021/acs.analchem.6b03279
- Tian H, Xu J, Xu Y, Guan Y. Multidimensional liquid chromatography system with an innovative solvent evaporation interface. Journal of Chromatography A. 2006;1137(1):42-48. Available from: https://doi.org/10.1016/j.chroma.2006.10.005
- Iguiniz M, Corbel E, Roques N, Heinisch S. Quantitative aspects in on-line comprehensive two-dimensional liquid chromatography for pharmaceutical applications. Talanta. 2019;195:272-280. Available from: https://doi.org/10.1016/j.talanta.2018.11.030
- Li JF, Yan X, Wu YL, Fang MJ, Wu Z, Qiu YK. Comprehensive two-dimensional normal-phase liquid chromatography × reversed-phase liquid chromatography for the analysis of toad skin. Analytica Chimica Acta. 2017;962:114-120. Available from: https://doi.org/10.1016/j.aca.2017.01.038
- Yang RJ, Wang N, Ma X, Gong MD, Wang YR, Meng SY, Liu ZY, et al. A novel two-dimensional liquid chromatography combined with ultraviolet detection method for quantitative determination of pyridoxal 5′-phosphate, 4-pyridoxine acid and pyridoxal in animal plasma. Animals. 2023;13(8):1333. Available from: https://doi.org/10.3390/ani13081333
- Goyon A, Dai L, Chen T, Wei B, Yang F, Andersen N, et al. From proof of concept to the routine use of an automated and robust multi-dimensional liquid chromatography-mass spectrometry workflow applied for the charge variant characterization of therapeutic antibodies. Journal of Chromatography A. 2020;1615:460740. Available from: https://doi.org/10.1016/j.chroma.2019.460740
- Lazzari E, Arena K, Caramão EB, Herrero M. Quantitative analysis of aqueous phases of bio-oils resulting from pyrolysis of different biomasses by two-dimensional comprehensive liquid chromatography. Journal of Chromatography A. 2019;1602:359-367. Available from: https://doi.org/10.1016/j.chroma.2019.06.016
- Bogdanova E, Pugajeva I, Reinholds I, Bartkevics V. Two-dimensional liquid chromatography-high resolution mass spectrometry method for simultaneous monitoring of 70 regulated and emerging mycotoxins in Pu-erh tea. Journal of Chromatography A. 2020;1622:461145. Available from: https://doi.org/10.1016/j.chroma.2020.461145
- Höhme L, Fischer C, Kleinschmidt T. Characterization of bitter peptides in casein hydrolysates using comprehensive two-dimensional liquid chromatography. Food Chemistry. 2023;404:134527. Available from: https://doi.org/10.1016/j.foodchem.2022.134527
- Arena K, Martín-Pozo L, Laganà Vinci R, Cacciola F, Dugo P, Mondello L. Determination of pesticide residues in five different corn-based products using a single and simple solid-liquid extraction without clean-up steps followed by comprehensive two-dimensional liquid chromatography coupled to tandem mass spectrometry. Microchemical Journal. 2024:111298. Available from: https://doi.org/10.1016/j.microc.2024.111298
- Martín-Pozo L, Arena K, Cacciola F, Dugo P, Mondello L. Development and validation of a multi-class analysis of pesticides in corn products by comprehensive two-dimensional liquid chromatography-tandem mass spectrometry. Journal of Chromatography A. 2023;1701:464064. Available from: https://doi.org/10.1016/j.chroma.2023.464064
- Wicht K, Baert M, Muller M, Bandini E, Schipperges S, von Doehren N, et al. Comprehensive two-dimensional temperature-responsive × reversed-phase liquid chromatography for the analysis of wine phenolics. Talanta. 2022;236:122889. Available from: https://doi.org/10.1016/j.talanta.2021.122889
- Reymond C, Masle AL, Colas C, Charon N. On-line two-dimensional liquid chromatography hyphenated to mass spectrometry and ion mobility-mass spectrometry for the separation of carbohydrates from lignocellulosic biomass. Journal of Chromatography A. 2021;1636:461716. Available from: https://doi.org/10.1016/j.chroma.2020.461716
- Armutcu C, Uzun L, Denizli A. Determination of Ochratoxin A traces in foodstuffs: Comparison of an automated on-line two-dimensional high-performance liquid chromatography and off-line immunoaffinity-high-performance liquid chromatography system. Journal of Chromatography A. 2018;1569:139-148. Available from: https://doi.org/10.1016/j.chroma.2018.07.057
- Grutzmann Arcari S, Arena K, Kolling J, Rocha P, Dugo P, Mondello L, et al. Polyphenolic compounds with biological activity in guabiroba fruits (Campomanesia xanthocarpa Berg.) by comprehensive two-dimensional liquid chromatography. Electrophoresis. 2020;41(20):1784-1792. Available from: https://doi.org/10.1002/elps.202000170
- Zelzer S, Meinitzer A, Enko D, Herrmann M, Goessler W. Development of a two-dimensional liquid chromatography-tandem mass spectrometry method for the determination of vitamin D2 in mushrooms. Anal Bioanal Chem. 2022;414(26):7565-7572. Available from: https://doi.org/10.1007/s00216-022-04281-3
- Bandini E, Wicht K, Barbetta MFS, Eghbali H, Lynen F. Temperature-responsive comprehensive two-dimensional liquid chromatography coupled to high-resolution mass spectrometry for the elucidation of the oxidative degradation processes of chemicals of environmental concern. J Chromatogr A. 2024;1719:464765. Available from: https://doi.org/10.1016/j.chroma.2024.464765
- de Souza AB, Ali I, van de Goor T, Dewil R, Cabooter D. Comprehensive two-dimensional liquid chromatography with high-resolution mass spectrometry to investigate the photoelectrochemical degradation of environmentally relevant pharmaceuticals and their degradation products in water. J Environ Manage. 2024;351:120023. Available from: https://doi.org/10.1016/j.jenvman.2024.120023
- Beschnitt A, Schwikowski M, Hoffmann T. Towards comprehensive non-target screening using heart-cut two-dimensional liquid chromatography for the analysis of organic atmospheric tracers in ice cores. J Chromatogr A. 2022;1661:462706. Available from: https://doi.org/10.1016/j.chroma.2021.462706
- Zhong J, Liu X, Chen L, Li K, Hu Q, Wu K, et al. Simultaneous separation and determination of several chiral antidepressants and their enantiomers in wastewater by online heart-cutting two-dimensional liquid chromatography. Ecotoxicol Environ Saf. 2023;263:115302. Available from: https://doi.org/10.1016/j.ecoenv.2023.115302
- Gachumi G, Schryer A, Siciliano SD. Two-dimensional ion chromatography tandem-mass spectrometric (2D-IC-MS/MS) method for the analysis of phosphorus compounds in soil. J Chromatogr A. 2024;:465287. Available from: https://doi.org/10.1016/j.chroma.2024.465287
- Purschke K, Zoell C, Leonhardt J, Weber M, Schmidt TC. Identification of unknowns in industrial wastewater using offline 2D chromatography and non-target screening. Sci Total Environ. 2020;706:135835. Available from: https://doi.org/10.1016/j.scitotenv.2019.135835
- Armutcu C, Özgür E, Karasu T, Bayram E. Rapid analysis of polycyclic aromatic hydrocarbons in water samples using an automated on-line two-dimensional liquid chromatography. Water Air Soil Pollut. 2019;230:1-11. Available from: https://doi.org/10.1007/s11270-019-4306-7
- Ouyang X, Leonards P, Legler J, van der Oost R, de Boer J, Lamoree M. Comprehensive two-dimensional liquid chromatography coupled to high-resolution time-of-flight mass spectrometry for chemical characterization of sewage treatment plant effluents. J Chromatogr A. 2015;1380:139-145. Available from: https://doi.org/10.1016/j.chroma.2014.12.075
- Pugajeva I, Ikkere LE, Jansons M, Perkons I, Sukajeva V, Bartkevics V. Two-dimensional liquid chromatography-mass spectrometry as an effective tool for assessing a wide range of pharmaceuticals and biomarkers in wastewater-based epidemiology studies. J Pharm Biomed Anal. 2021;205:114295. Available from: https://doi.org/10.1016/j.jpba.2021.114295
- Zhu K, Pursch M, Eeltink S, Desmet G. Maximizing two-dimensional liquid chromatography peak capacity for the separation of complex industrial samples. J Chromatogr A. 2020;1609:460457. Available from: https://doi.org/10.1016/j.chroma.2019.460457
- Stoll D, Sylvester M, Meston D, Sorensen M, Maloney TD. Development of multiple heartcutting two-dimensional liquid chromatography with ion-pairing reversed-phase separations in both dimensions for analysis of impurities in therapeutic oligonucleotides. J Chromatogr A. 2024;1714:464574. Available from: https://doi.org/10.1016/j.chroma.2023.464574
- Lin Z, Wang Q, Zhou Y, Shackman JG. Trapping mode two-dimensional liquid chromatography for quantitative low-level impurity enrichment in pharmaceutical development. J Chromatogr A. 2023;1700:464043. Available from: https://doi.org/10.1016/j.chroma.2023.464043
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.



 Save to Mendeley
Save to Mendeley
