Open Journal of Analytical and Bioanalytical Chemistry
Benzofuran and Naphthofuran based chemosensors for metal ion detection using fluoroscence spectroscopy
Department of Chemistry and Center of Advanced Studies, Panjab Univesity, Chandigarh, PIN 160014, India
Author and article information
Cite this as
Ali S, Joshi S, Agarwal J (2022) Benzofuran and Naphthofuran based chemosensors for metal ion detection using fluoroscence spectroscopy. Open J Anal Bioanal Chem. 2022; 6(1): 18-23. Available from: 10.17352/ojabc.000026
Copyright License
© 2022 Ali S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.With the advancement in the field of agriculture and industrial regime, numerous metals such as lead, cadmium, mercury, zinc, copper, arsenic, etc. are released into the environment as effluent. These metal ions enter water bodies and generate many health issues. Considering their harmful impact on human lives, numerous fluorescent probes have been developed in recent years to detect the presence of these metal ions. The fluorescent probes are used owing to their ease of operation, good selectivity, and high sensitivity. The interaction between functional groups present in the structural framework of the probe and the empty orbitals of metal ions is responsible for the chemo selectivity of the probe. This report summarizes the organic probes which possess benzofuran and naphthofuran moieties and act as fluorescent chemosensors toward metal ions. The selectivity and sensitivity of these chemosensors along with the limit of detection have also been discussed.
Metal ions play a significant role in our biological system. Every metal ion has its function to perform various biological and metabolic activities. All these metal ions are categorized as essential and non-essential metals [1]. The human body largely constitutes essential metal ions (Na+, K+, Ca2+, Mg2+) often known as bulk elements while other metals such as Mn, Fe, Co, Cu, Zn, and Mo are present in trace amounts and are indispensable for humans life. Although metal ions are necessary for different biological processes, an appropriate range of concentration should be maintained in organisms. Both deficiency and excess of these metal ions can cause serious ailments in the human body [2,3]. Nonessential metals or heavy metals (Cd, Pb, Hg, As, Cr, Ni, Al, Tl) are a threat to human life due to their toxic nature and non-biodegradability [4].
The World Health Organization (WHO) and Environmental Protection Agency (EPA) have strictly defined concentration limits of these metal ions in the human body [5,6]. Therefore, it becomes necessary to detect these metal ions at an early stage. For that purpose, various techniques have been developed to detect these metal ions like FAAS [7], AAS [8], ICP-MS [9], inductively coupled plasma emission spectrometry [10], and voltammetry [11]. However, the complicated instrumentation, skilled manpower, and costly operation have restricted their utilization in metal ion detection. As an alternative to these traditional methods, fluorescence spectroscopy is widely used due to its high selectivity and sensitivity, low cost, immediate response, easy visualization, and simple operational management [12]. As a result, several fluorescent probes known as chemosensors have been synthesized and explored for metal ion detection.
Chemosensors
Chemosensors are compounds that bear a binding site and define a mechanism of interaction between the analyte and the fluorophore. In the past few decades, diverse fluorescent chemosensors have been developed for metal ions detection [13,14]. The first fluorescent chemosensor was developed in 1867, by Goppelsroder, et al. This group reported a morin ring forming a strongly fluorescent chelate with Al3+ [15]. A chemosensing system generally consisted of a receptor and an active unit (chromophore/fluorophore) which can modify its properties upon interactions with the analyte and can react to the system by a “turn-on-off” mechanism in the presence of metal ions [16]. Most of the chemosensors which detect metal ions via fluorescence are generally based on the acceptor-fluorophore system, where the acceptor binds or chelates to the metal ion and the fluorophore translates this binding to fluorescence signal. Various moieties such as rhodamine dyes, amides, naphthalimides, pyrenes, azamacrocycles, Schiff bases, furan, and salicylaldehyde could serve as binding sites and have been used for the detection of metal ions [17-19].
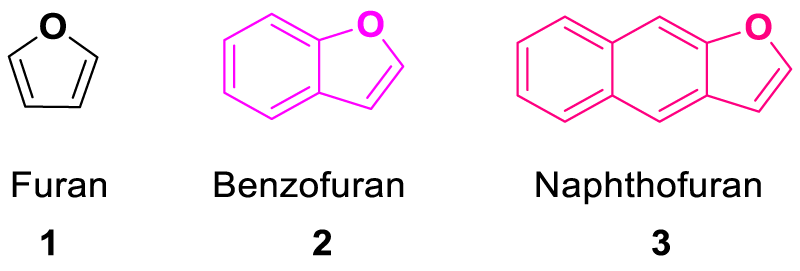
Benzofuran and naphthofuran molecules are the ring-fused furan derivatives (Figure 1). These moieties are expected to be fluorescent in nature due to the presence of a highly conjugated and electron-rich system. The presence of oxygen atoms and extended π-system in the aromatic skeleton of the benzofuran and naphthofuran molecules make them electron-rich molecules and such molecules show significant interaction with the electron-deficient metal ions. Naphthofuran molecules have a large aromatic system as compared to benzofuran molecules and possess better absorption and emission spectra. Many reports are available in the literature having benzo- and naphthofuran-derived molecules, which are fluorescent in nature and exhibit chemoselectivity for metal ion detection. However, to the best of our knowledge, no review is available in the literature listing such molecules and showcasing their significance in designing novel chemosensors and in the effective interaction of probes with electron-deficient metal ions. Thus, in this review, we have reported such benzo- and naphthofuran-based fluorescent chemosensors and discussed their interaction mechanism and selectivity for metal ion detection.
Benzofuran based chemosensors
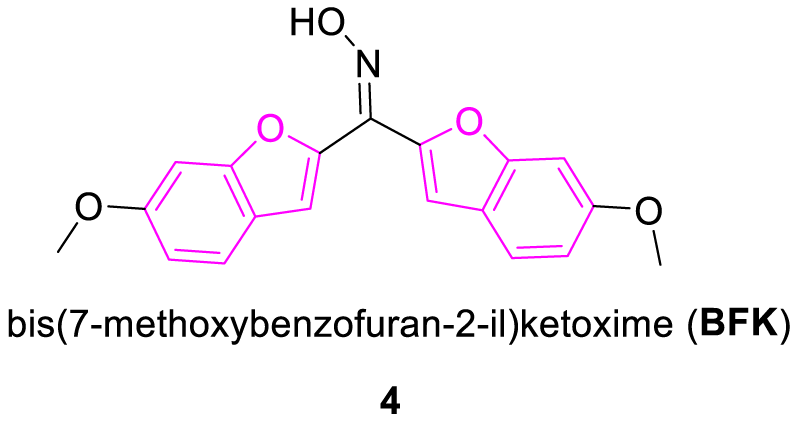
Oter, et al. in 2007 synthesized a benzofuran-based fluorescent dye molecule (BFK, 4) which was explored as a sensing agent in the optical sensor design. The photoluminescence property of this molecule was tested in various solvents along with the polymer matrix. In the polyvinylchloride (PVC) matrix, it displayed a large Stoke shift value which allowed the emitted fluorescence photons to be easily distinguished from the excitation photons. It led to the generation of a very low background signal and permitted the use of BFK dye in the construction of fiber optic sensors. This PVC-based optical sensor was prepared from PVC, plasticizer, BFK dye, potassium tetrakis (4-chlorophenyl) borate and THF, which was explored as a sensing film. This dye-doped membrane showed remarkable quenching toward the Fe3+ ion as BFK dye is supposed to form a non-fluorescent complex with Fe3+. The selective quenching of Fe3+ ion by membrane was mainly accountable to the person’s Hard Soft Acid Base (HSAB) principle which states that hard acids prefer to bind with a hard base and soft acids prefer to bind with a soft base. The O- and N- donor atoms on the BFK dye were in the category of hard Lewis base which was probably responsible for binding with Fe3+. As an optic sensor, it can be used as an alternative to fluorescent Fe3+ indicators excited with UV light (Figure 2) [20].
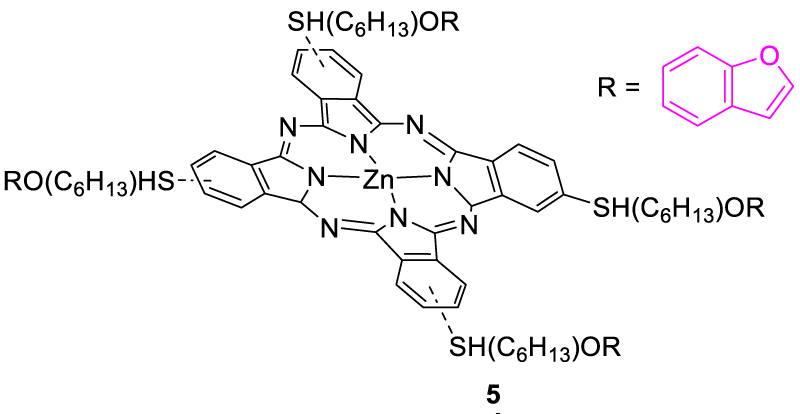
In 2009, Kandaz, et al. synthesized fluorescent chemosensor 2,9,16,23-tetrakis-{6-(-benzofuran-2-carboxylate)-hexylthio}-phthalocyanine Zinc (II) (ZnPcBzF, 5), which showed distinct changes in absorbance and fluorescence spectra upon treatment with silver ion Ag(I) solution. Upon excitation at 616 nm, the fluorescence emission of ZnPcBzF (5) was decreased with the addition of silver ions. It indicated the formation of the silver ion complex via the sulfur atom on the sensor, which later altered the fluorescence emission of the phthalocyanine (Pc) core. The reason for the quenching of fluorescence emission was probably the intermolecular aggregation induced by silver ion which brought phthalocyanine units close enough to each other and interacted with the π-electrons of the Pc core (Figure 3) [21].
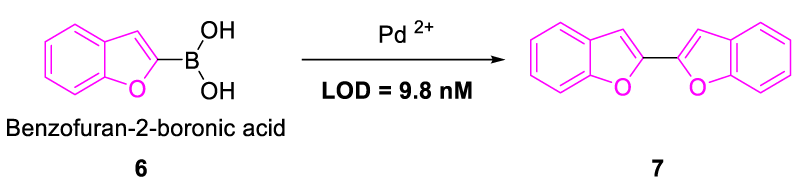
In 2017, Higashi, et al. synthesized Benzofuran-2-boronic acid (6), which was explored as an effective chemosensor for the detection of Pd2+. It showed a significant rise in the emission spectra upon treatment with Pd2+, giving a “turn-on” response. High selectivity for Pd2+ was displayed even in the presence of other competitive metal ions with a low detection limit of 9.8 nM. Upon addition of Pd2+ to commercially available benzofuran-2-boronic acid, it got converted into a highly fluorescent benzofuran dimer (7) under basic conditions, thus acting as a potential chemosensor for the detection of Pd2+ ions (Figure 4) [22].
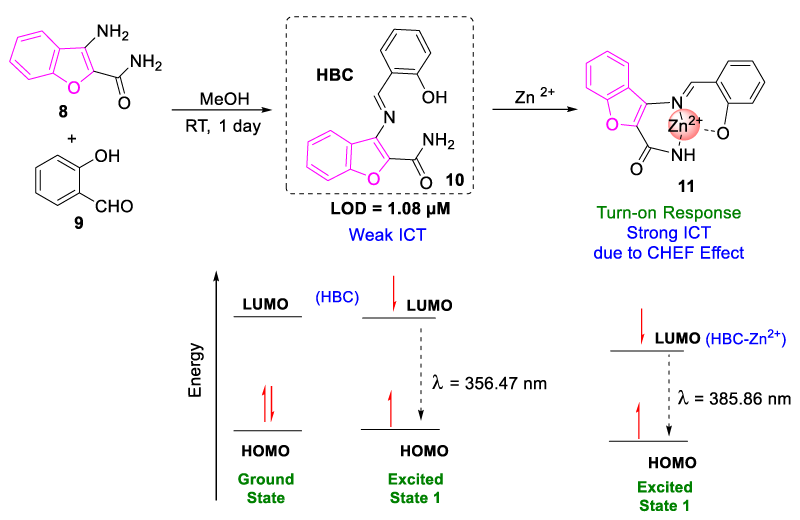
In 2018, Jang, et al. synthesized a benzofuran functionalized, ((E)-3-((2-hydroxybenzylidene)-amino)-benzofuran-2-carboxamide) chemosensor (HBC, 10) by the condensation reaction of 3-aminobenzofuran-2-carboxamide (8) and salicylaldehyde (9) for the selective detection of Zn2+. No interference of other ions was observed in the turn-on spectra of the HBC-Zn2+ complex and its detection limit was found to be 1.08 µM. From the theoretical analysis, it was observed that the first transition in probe 10 occurred from HOMO to LUMO due to the intramolecular charge (ICT) transition. When Zn2+ metal ion interacted with probe 10 it decreased the energy difference between HOMO-LUMO orbitals due to the chelation-enhanced-fluorescence (CHEF) effect. It reduced the non-radiative transition and resulted in the HBC-Zn2+ complex exhibiting turn-on spectra (Figure 5). The recyclability of probe HBC with Zn2+ using EDTA was also established which gave reversible fluorescence variation upon alternate successive addition of Zn2+ and EDTA [23].
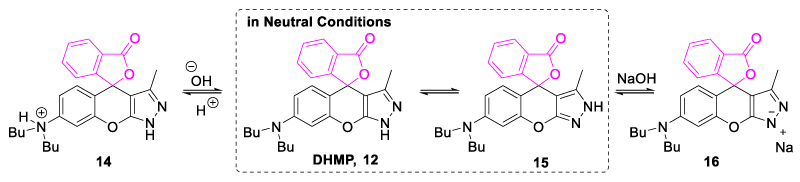
In 2019, Yue, et al. synthesized and developed {7-(dibutylamino)-3-methyl-1H, 3´H-spiro [chromeno-[2,3-c]-pyrazole-4,1´-[2]-benzofuran]-3´-one (DHMP, 12) as highly selective chemosensor toward the copper (II) ion sensing in an aqueous medium (Figure 6). DHMP probe was found to be pH dependent as its selectivity toward copper metal was due to the protonation of the nitrogen atom (Figure 7). In a neutral medium, equilibrium occurred between two forms 12 and 15; but in an acidic medium, the DHMP molecule got protonated at the dibutylamino nitrogen atom (14), and in the basic medium deprotonation of pyrazole ring occurred to form sodium salt (16) (Figure 7). Chemosensor showed selectivity under strongly acidic conditions (pH range 1.1-2.1) and strong alkaline conditions (pH range 13.1-13.9). No significant emission intensity was found in the pH range of 2.9 to 11.9. Thus, the chemoselectivity of probe DHMP was observed to be strongly pH-dependent due to the protonation and deprotonation mechanism of dibutyl amino nitrogen and pyrazole ring [24].
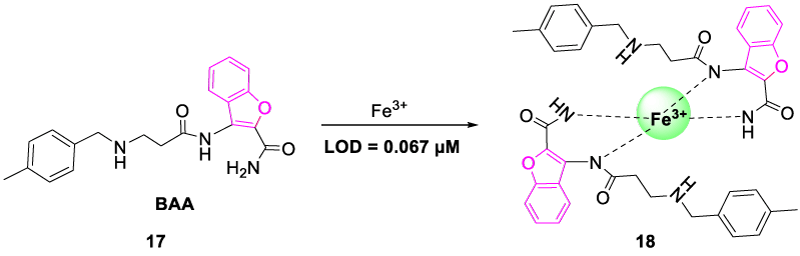
In 2019, Madhu, et al. synthesized a benzofuran-based chemosensor 3-(3-((4-methylbenzyl)-amino)-propanamide)-benzofuran-2-carboxamide (BAA, 17) which showed a ‘turn-on’ response with high selectivity and sensitivity towards Fe3+ ions in DMSO/H2O solution (9:1). Due to the paramagnetic nature of Fe3+, most of the reported iron sensors showed a ‘switch-off response. Probe BAA showed an intramolecular charge transfer process in the Free State which was evident via theoretical analysis of HOMO and LUMO orbitals performed by the authors. Upon the addition of Fe3+ ion, the ICT process was diminished, and a strong CHEF effect was observed which displayed turn-on spectra. In addition to this, the HOMO-LUMO gap was dropped from 4.02 eV in the case of BAA to 1.62 eV in the case of [BAA]2-Fe3+ complex which confirmed the strong binding of sensor BAA with Fe3+ and stable complex formation. Furthermore, chemosensor BAA showed ‘ON-OFF-ON’ fluorescence signals upon alternate addition of Fe3+and EDTA after several cycles. Upon the addition of EDTA, the decrease in fluorescence intensity was observed due to the formation of a strong complex between EDTA and Fe3+, which further suggested that the sensor BAA could be used as a reversible chemosensor for Fe3+ (Figure 8) [25].
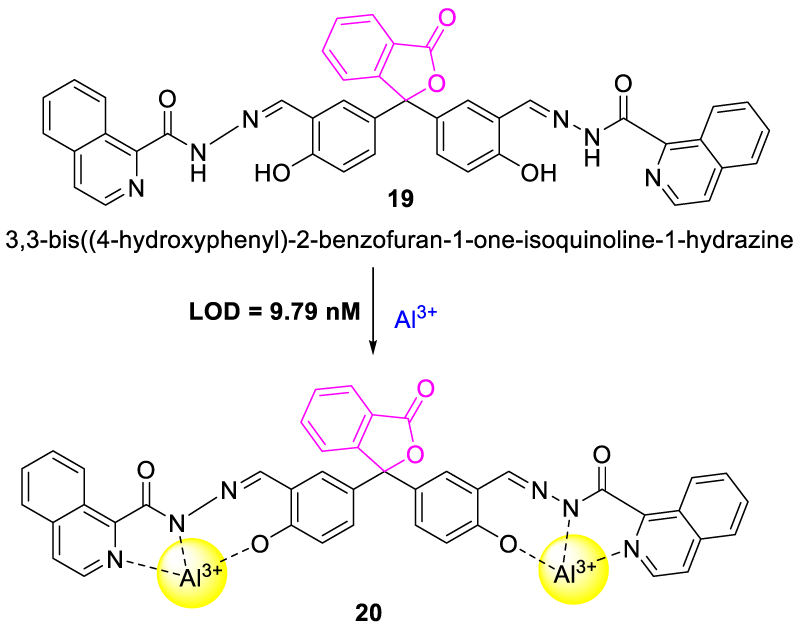
In 2020, Zhu, et al. synthesized Schiff base [3,3-bis((4-hydroxyphenyl)-2-benzofuran-1-one-isoquinoline-1-hydrazine] (19) from phenolphthalein and isoquinoline hydrazide. Upon excitation at 377 nm, probe 19 displayed an extremely weak fluorescence emission profile (emission at 479 nm) with a very low quantum yield value of 0.0012. This was caused by the lone pair electrons of the nitrogen atom in the Schiff base on reaching the chromone moiety which suppressed the fluorescence via the photoinduced electron transfer (PET) effect. But upon the addition of Al3+, the fluorescent intensity of probe 19 was increased gradually up to 700-fold and the quantum yield reached 0.032 due to the generation of the CHEF effect that diminished the PET effect [26]. Thus, the chemosensor showed distinguished spectra with Al3+ but no other metal showed an interference effect except Cu2+ metal which quenched the fluorescence of 19 due to its paramagnetic nature Figure 9.
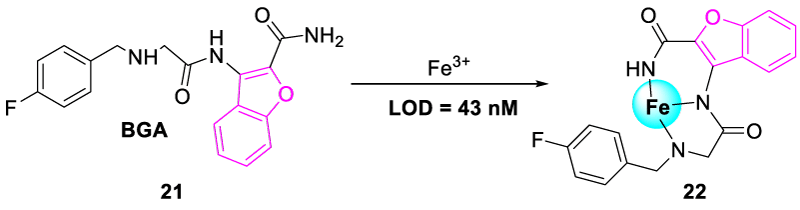
In 2022, Madhu, et al, synthesized benzofuran glycinamide-based chemosensor, 3-(2-([4-fluorobenzyl]-amino)-acetamido)-benzofuran-2-carboxamide (BGA, 21) via multistep reaction for the selective and sensitive detection of Fe3+ ions. Chemosensor BGA showed a “turn-off” fluorescence response towards Fe3+ ion even in the presence of other metal ions. Probe BGA had a large electron density on the glycinamide moiety instead of the benzofuran scaffold in the HOMO orbital and possessed intramolecular charge transfer spectra (ICT). When Fe3+ ion interacted with BGA, the energy level of LUMO decreased which changed the photo behavior of the probe BGA. Due to this phenomenon, the ICT process was diminished, and a strong chelation effect occurred causing the quenching of BGA fluorescence. The reusability of probe BGA was further accepted by repeated alternate addition of EDTA and Fe3+ giving “turn off-on-off” spectra. Fe3+ responsive chemosensor BGA showed high selectivity and sensitivity with a very low detection limit of 43 nM (Figure 10) [27].
Naphthofuran based chemosensors
Though several naphthofuran-based molecules have been synthesized and utilized in different fields such as medicinal chemistry, catalysis, semiconducting devices, etc. [28-35]; only sporadic reports are available in literature exploring such molecules in the chemosensing field using fluorescence spectroscopy.
In 2015, Jie, et al. synthesized naphthofuran carbonyl hydrazone derived Zn2+ coordinated complex (23) as a fluorescent chemosensor via a displacement approach. Upon treatment with Cu2+, the chemosensor exhibited an excellent “turn-off” response. To know the binding mode of Cu2+ to probe 23, an NMR measurement experiment had been carried out which showed the disappearance of the -OH (ẟ 12.22) and -NH (ẟ 11.24) protons in the NMR spectra of probe 23. When Cu2+ ion interacted with probe 23, displacement of Zn2+ occurred, and turn-off spectra were observed due to the paramagnetic properties of Cu2+. The chemosensor efficiently detected Cu2+ even in the presence of other metal ions with a detection limit of 2.3 x 10-7 M (Figure 11) [36].
In 2022, Guan, et al. synthesized a naphthofuran functionalized naphthalimide (25) chemosensor for the selective detection of highly toxic Hg2+ ions. The chemosensor 25 showed a “turn-off” response for Hg2+ ions with a low detection limit of 4.7 x 10-7 M, displaying the excellent stimuli-responsive property for Hg2+ in the presence of other metal ions. From the theoretical analysis of the HOMO-LUMO orbital and electron potential map of 25 and 26, it was concluded that the turn-off spectra were observed due to the charge transfer between 25 and Hg2+ (Figure 12) [37].
Conclusion and future prospective
This report summarizes the organic molecules which possess benzofuran and naphthofuran moieties in their structural framework and act as efficient chemosensors toward metal ion detection. The presence of benzofuran and naphthofuran moieties make organic molecules electron rich that can interact with an empty orbital of metal ion effectively and produce significant emission spectra. The performance of these chemosensors has been listed in Table 1 along with their limit of detection.
Most of the listed chemosensors were found to be highly sensitive as LOD was achieved up to 9.79 nM. All the probes were found to be highly selective towards one specific metal ion. Some of them such as probes 10, 17, and 21 were able to show reusability due to their reversible interaction with metal ions. Thus the studies clearly depict the potential of these molecules in the chemosensing field. Unfortunately, not many chemosensors have been designed keeping in mind the structural benefits of benzofuran and naphthofuran skeleton. In this scenario, we believe that this article may give an opportunity to the chemists to design and develop novel benzo- and naphthofuran-based chemosensors which can be explored in near future more effectively for the detection of various metals.
We are grateful to Science and Engineering Research Board, Delhi (Grant no(s). CRG/2021/006506 and EMR/2017/000453), India for providing financial assistance. S.A. thanks UGC, New Delhi for the award of a research fellowship (F. No. 61-1/2019 (SA-III)).
- Crichton RR, Ward RJ, Hider RC. Metal Toxicity- An Introduction. In Metal Chelation in medicine. Metallobiology Series No. 8: RSC, 2016;1-23.
- Jaishankar M, Tseten T, Anbalagan N, Mathew BB, Beeregowda KN. Toxicity, mechanism and health effects of some heavy metals. Interdiscip Toxicol. 2014 Jun;7(2):60-72. doi: 10.2478/intox-2014-0009. Epub 2014 Nov 15. PMID: 26109881; PMCID: PMC4427717.
- Balali-Mood M, Naseri K, Tahergorabi Z, Khazdair MR, Sadeghi M. Toxic Mechanisms of Five Heavy Metals: Mercury, Lead, Chromium, Cadmium, and Arsenic. Front Pharmacol. 2021 Apr 13;12:643972. doi: 10.3389/fphar.2021.643972. PMID: 33927623; PMCID: PMC8078867.
- Jia K, Yi Y, Ma W, Cao Y, Li G, Liu S, Wang T, An N. Ion flotation of heavy metal ions by using biodegradable biosurfactant as collector: Application and removal mechanism. Miner Eng. 2022; 176: 107338.
- Kinuthia GK, Ngure V, Beti D, Lugalia R, Wangila A, Kamau L. Levels of heavy metals in wastewater and soil samples from open drainage channels in Nairobi, Kenya: community health implication. Sci Rep. 2020 May 21;10(1):8434. doi: 10.1038/s41598-020-65359-5. Erratum in: Sci Rep. 2020 Jul 7;10(1):11439. PMID: 32439896; PMCID: PMC7242368.
- Saha SK, Ghosh KR, Hao W,Wang ZY, Ma J, Chiniforooshan Y, Bock WJ. Highly sensitive and selective fluorescence turn-on detection of lead ion in water using fluorene-based compound and polymer. J Mater Chem A. 2014; 2: 5024.
- Ipeaiyeda AR, Ayode AR. Flame atomic absorption spectrometric determination of heavy metals in aqueous solution and surface water preceded by co-precipitation procedure with copper (II) 8-hydroxyquinoline. Appl Water Sci. 2017; 7: 4449-4459.
- Akram S, Najam R, Rizwani GH, Abbas SA. Determination of heavy metal contents by atomic absorption spectroscopy (AAS) in some medicinal plants from Pakistani and Malaysian origin. Pak J Pharm Sci. 2015 Sep;28(5):1781-7. PMID: 26408897.
- Dressler VL, Pozebon D, Curtius A. Determination of heavy metals by inductively coupled plasma mass spectrometry after on-line separation and preconcentration, Spectrochim. Acta Part B at Spectroscopsc. 1998; 1527-1539.
- Wu J, Yu J, Li J, Wang J, Ying Y. Detection of metal ions by atomic emission spectroscopy from liquid-electrode discharge plasma, Spectrochim. Acta Part B at Spectroscopsc. 2007; 62: 1269-1272.
- Ding R, Cheong YH, Ahamed A, Lisak G. Heavy Metals Detection with Paper-Based Electrochemical Sensors. Anal Chem. 2021 Feb 2;93(4):1880-1888. doi: 10.1021/acs.analchem.0c04247. Epub 2021 Jan 12. PMID: 33430590.
- Kumawat LK, Kumar M, Bhatt P, Sharma A, Asif M, Gupta VK. An easily accesible optical chemosensor for Cu 2+ based on novel imidazoazine framework,its performance characteristics and potential applications. Sens. Actuators B: Chem. 2017; 240: 365-375.
- Wang Y, Song F, Zhu J, Zhang Y, Du L, Kan C. Highly selective fluorescent probe based on a rhodamine B and furan-2-carbonyl chloride conjugate for detection of Fe 3+ in cells. Tetrahedron Lett. 2018; 59: 3756-3762.
- Peng H, Liu Y, Huang J, Huang S, Cai X, Xu S, Huang A,Zeng Q, Xu M. A simple fluorescent probe for selective detection of Al 3+ based on furan Schiff base and its crystal structures. J. Mol. Struct. 2021; 1229: 129866.
- Wu D, Sedgwick AC, Gunnlaugsson T, Akkaya EU, Yoon J, James TD. Fluorescent chemosensors: the past, present and future. Chem Soc Rev. 2017 Dec 7;46(23):7105-7123. doi: 10.1039/c7cs00240h. Epub 2017 Oct 11. PMID: 29019488.
- Farhi A, Firdaus F, Saeed H, Mujeeb A, Shakir M, Owais M. A quinoline-based fluorescent probe for selective detection and real-time monitoring of copper ions - a differential colorimetric approach. Photochem Photobiol Sci. 2019 Dec 1;18(12):3008-3015. doi: 10.1039/c9pp00247b. Epub 2019 Nov 7. PMID: 31696896.
- Jang HK, Kim A, Jung JM, Lee M, Lim HM, Kim C. Detection of Zinc(II) by a fluorescence chemosensor based on Benzofuran in Aqueous media and live cells. Chem. Soc. 2018; 39:1373-1379.
- Sarkar D, Pramanik AK, Mondal TK. Coumarin based dual switching fluorescent ‘turn-on’ chemosensor for selective detection of Zn 2+ and HSO4ˉ: an experimental and theoretical study. RSC Adv. 2014; 4:25341.
- Bai L, Xu Y, Li L, Tao F, Wang S, Wang L, Li G. An efficient water-soluble fluorescent chemosensor based on furan Schiff base functionalised PEG for thr sensitive detection of Al 3+ in pure aqueous solution. New J. Chem. 2020;44: 11148.
- Oter O, Ertekin K, Kirilmis C, Koca M, Ahmedzade M. Characterization of a newly synthesized fluorescent benzofuran derivative and usage as a selective fibre optic sensor for Fe (III), Sens. Actuators B. Chem. 2007;122: 450-456.
- Kandaz M, Guney O, Senkal FB. FluroscentChemosensor for Ag(I) based on amplified fluorescence quenching of a new phthocyanine bearing derivative of benzofuran. Polyhedron. 2009; 28: 3110-3114.
- Higashi A, Kishikawa N, Ohyama K, Kuroda N. A simple and highly selective fluorescent sensor for palladium based on benzofuran-2-boronic acid. Tetrahedron Lett. 2017; 58: 2774-2778.
- Jang HK,Kim A, Jung JM, Lee M, Lim HM, Kim C. Detection of Zinc(II) by a fluorescence chemosensor based on Benzofuran in Aqueous media and live cells. Chem. Soc. 2018; 39: 1373-1379
- Yue Y; Xie Z; Chu Y, Shi W. A novel fluorescent probe based on spiro [chromeno[2,3-c]-pyrazole-4,1’-[2]-benzofuran]-3’-one for detecting Copper(II) ions in aqueous solution. Russ. J. Gen. Chem. 2019; 55(6): 866-873.
- Madhu P, Sivakumar P, Sribalan R. A benzofuran-β-alaninamide based “turn-on” fluorescent chemosensor for selective recognition of Fe 3+ ions. New J. Chem. 2019; 43: 14426.
- Zhu J, Lu L, Wang M, Sun T, Huang Y, Wang C, Bao W, Wang M, Zou F,Tang Y. Fluorescence “On-Off” chemical sensor for ultrasensitive detection of Al 3+ in live cell. Tetrahedron Lett. 2020; 61: 151893.
- Madhu P, Sivakumar P, Sribalan R, Arumugam SM. Highly selective and sensitive 'on-off' fluorescent chemosensor for Fe 3+ ions crafted by benzofuran moiety in both experimental and theoretical methods. Luminescence. 2022 Jul;37(7):1064-1072. doi: 10.1002/bio.4258. Epub 2022 May 4. PMID: 35434912.
- Knudsen Dal NJ, Speth M, Johann K, Barz M, Beauvineau C, Wohlmann J, Fenaroli F, Gicquel B, Griffiths G, Alonso-Rodriguez N. The zebrafish embryo as an in vivo model for screening nanoparticle-formulated lipophilic anti-tuberculosis compounds. Dis Model Mech. 2022 Jan 1;15(1):dmm049147. doi: 10.1242/dmm.049147. Epub 2022 Jan 26. PMID: 34842273; PMCID: PMC8807572.
- Kumar A, Srivastava G, Srivastava S, Verma S, Negi AS, Sharma A. Investigation of naphthofuran moiety as potential dual inhibitor against BACE-1 and GSK-3β: molecular dynamics simulations, binding energy, and network analysis to identify first-in-class dual inhibitors against Alzheimer's disease. J Mol Model. 2017 Aug;23(8):239. doi: 10.1007/s00894-017-3396-7. Epub 2017 Jul 24. PMID: 28741112.
- Rani D, Gulati V, Guleria M, Singh SP, Agarwal J. Aqueous synthesis of 2-Aryl -3-nitro -2H-chromenes via L-prolinamide mediated tandem oxa-Michael Henry reactions. J. Mol. Struct. 2022; 1265: 133341.
- Rani D, Sethi A, Kaur K, Agarwal J. Ultrasonication-Assisted Synthesis of a d-Glucosamine-Based β-CD Inclusion Complex and Its Application as an Aqueous Heterogeneous Organocatalytic System. J Org Chem. 2020 Aug 7;85(15):9548-9557. doi: 10.1021/acs.joc.0c00420. Epub 2020 Jul 27. PMID: 32672959.
- Rani D, Bhargava M, Agarwal J. Asymmetric Michael Addition of Unactivated Ketones with β-Nitrostyrenes Mediated by Bifunctional L-Prolinamide Organocatalysts. Chemistry Select. 2020; 5: 2435-2440.
- Rani D, Thakur L, Khera M, Goel N, Agarwal J. Mechanistic details of amino acid catalyzed two-component Mannich reaction: experimental study backed by density functional calculations. Amino Acids. 2019 Nov;51(10-12):1609-1621. doi: 10.1007/s00726-019-02798-z. Epub 2019 Nov 11. PMID: 31712920.
- Sousa C, Polonia M, Coelho AJ. Photochromic hybrid material doped with vinylidene-naphthofurans, Prog. Org. Coat. 2018; 125: 146-152.
- Zheng B, Huo L. Recent Advances of Furan and Its Derivatives Based Semiconductor Materials for Organic Photovoltaics. Small Methods. 2021 Sep;5(9):e2100493. doi: 10.1002/smtd.202100493. Epub 2021 Aug 16. PMID: 34928062.
- Jie G, Peng Z, Taibao W, Qi L, Hong Y, Youming Z. A highly selective fluorescent Chemosensor for Cu 2+ . Chem. Res. Chin. Univ. 2015; 31: 347-351.
- Guan W, Zhang Y, Zhang Q, Zhang Y, Wei T, Yao H. A novel fluorescent chemosensor based on naphthofuran functionalized naphthalimide for highly selective and sensitive detecting Hg 2+ and CN-. J Lumin. 2022; 244: 118722.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.
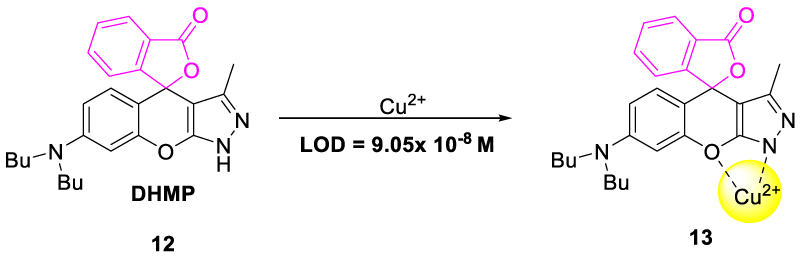


 Save to Mendeley
Save to Mendeley
