Open Journal of Analytical and Bioanalytical Chemistry
Recent advancements in fluoroquinolones removal techniques from wastewater
Teaching and Research Assistant, Department of Chemistry, University of Bahrain, Bahrain
Author and article information
Cite this as
Saqer H (2024) Recent advancements in fluoroquinolones removal techniques from wastewater. Open J Anal Bioanal Chem. 2024; 8(1): 7-16. Available from: 10.17352/ojabc.000031
Copyright License
© 2024 Saqer H. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Fluoroquinolones (FQs) are widely used antibiotics for therapeutic purposes, yet their extensive usage and unwise disposal led to antibiotic resistance as well as their widespread across the environment and becoming a major contaminates in water bodies as the wastewater treatment plants are not capable removing of FQs completely. Knowing the latter, the researchers endeavored to find techniques for the detection and removal of such contaminates from the disposed water from different facilities. This review highlights some of the recently and currently developed techniques for detection and removal. It discusses also different adsorbents used to remove FQs from wastewater. All in all, the removal of such leading contaminates from water would inhibit their spread across different environmental spheres thus promoting better environmental health.
Human beings and animals need pharmaceutical drugs as an essential part of maintaining their health and well-being as they are used for therapeutic ends. Such drugs are biologically active as well as they can persist in the environment due to their stability. Due to the latter, they are considered emerging pollutants. The toxicity and reactivity of such pollutants are mostly left untouched in terms of the permissible levels in which they could be disposed to the environment [1]. Such an issue induced researchers to proceed towards inventing new solutions for the emerging problem in the environment.
Among the many drugs used to cure illnesses and sicknesses, antibiotics are the most frequently used to combat microbial activity within bodies. Even though these drugs are useful for the latter purpose, they have introduced a problem that is lurking at the bay. Nowadays, antibiotics are classified as a dreadful threat to human beings, animals, and in short the environment as a whole [2]. Upon the discharge of antibiotics to the environment, usually, they are still posing activity since this, microorganisms protect themselves by developing resistance towards such drugs and thus the used drugs become deactivated therefore are not capable of achieving their ultimate aim. The previous phenomenon since it became a viral problem, was designated a term: Multidrug Resistance (MDR) [3]. MDR resulted in the development of uncurable or hardly curable infections with conventional anti-biotic agents for Gram-positive and Gram-negative bacteria [4].
Many sources are considered as an entrance of antibiotics to the environment, yet hospital discharge or effluent is known to be the most contributing source of such pollutants. Patients once intake as a part of their therapeutic process, such drugs get excreted and urinated by them and thus enter into the aquatic system due to the improper treatment process of the generated wastewater. The characteristics of hospital effluent are varying parameters depending upon many factors such as country, density of beds, healthcare facilities, wards, the used anti-biotics… etc. [5].
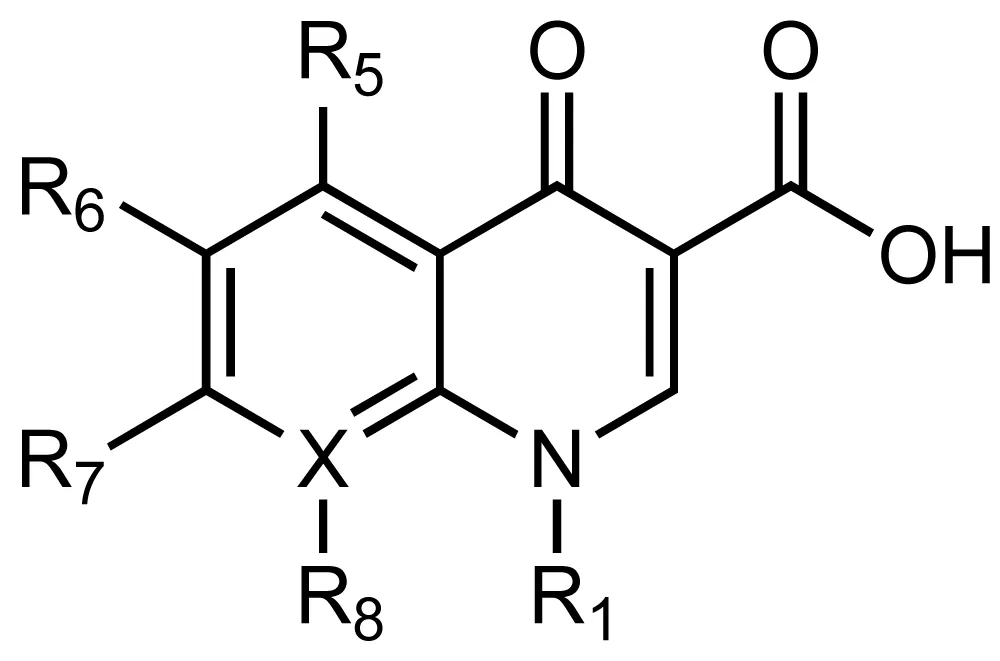
Just between the years 1930 and 1962, the world had produced more than 20 novel classes of antibiotics. Many of these were marketed and others were withdrawn. Classes like Penicillins, Cephalosporins, Aminoglycosides, Tetracyclines, Glycopeptides, and Quinolones are widely used and discharged into the environment [6]. Quinolones are a class of antibiotics characterized by the presence of bicyclic core as Figure 1 shows. Antibiotics have been used extensively to treat severe infection cases since their discovery during the early 1960s [7].
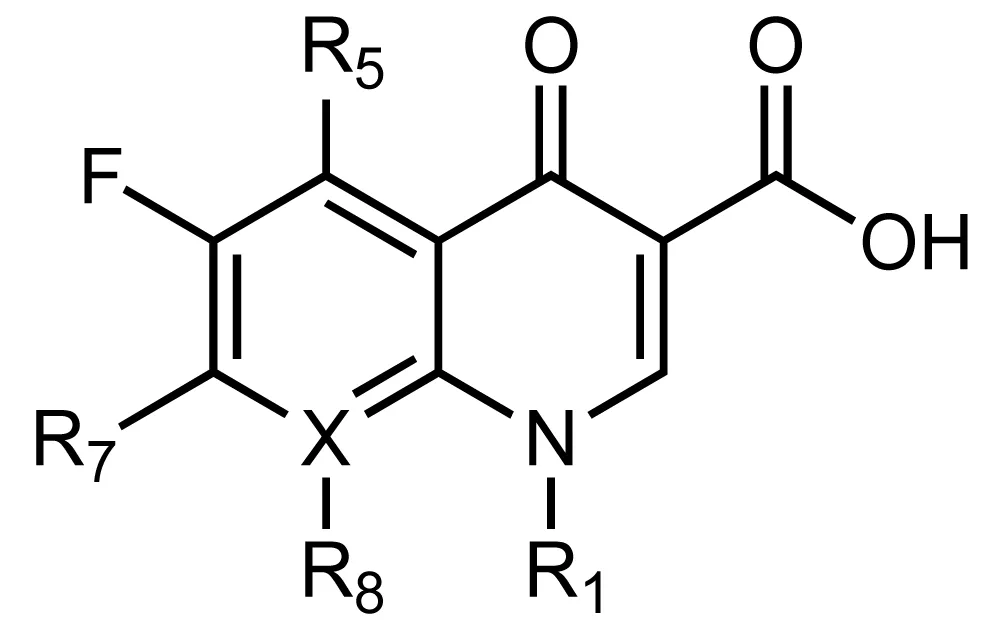
Since the discovery of nalidixic acid which is used to treat uncomplicated urinary tract infections., scientists devoted more efforts hoping to elevate its potential as it was observed that it is of great potency. The discovery of 6-fluoroquinolone, which is a nalidixic acid derivative, was the breakthrough towards further advancements and the discovery of the potential lies within Fluoroquinolones (FQs). Compared to the first generation of quinolones, fluoroquinolones are much more active as they could be used for Gram-positive and Gram-negative pathogens along with the latter, they are also active causative agents of tuberculosis, Mycobacterium tuberculosis. FQs, compared to the normal quinolones contain a fluoro-group at position R6 as Figure 2 shows, which indeed enhanced the Gram-negative activity, and since then, all the quinolones were designed as fluoroquinolones [7].
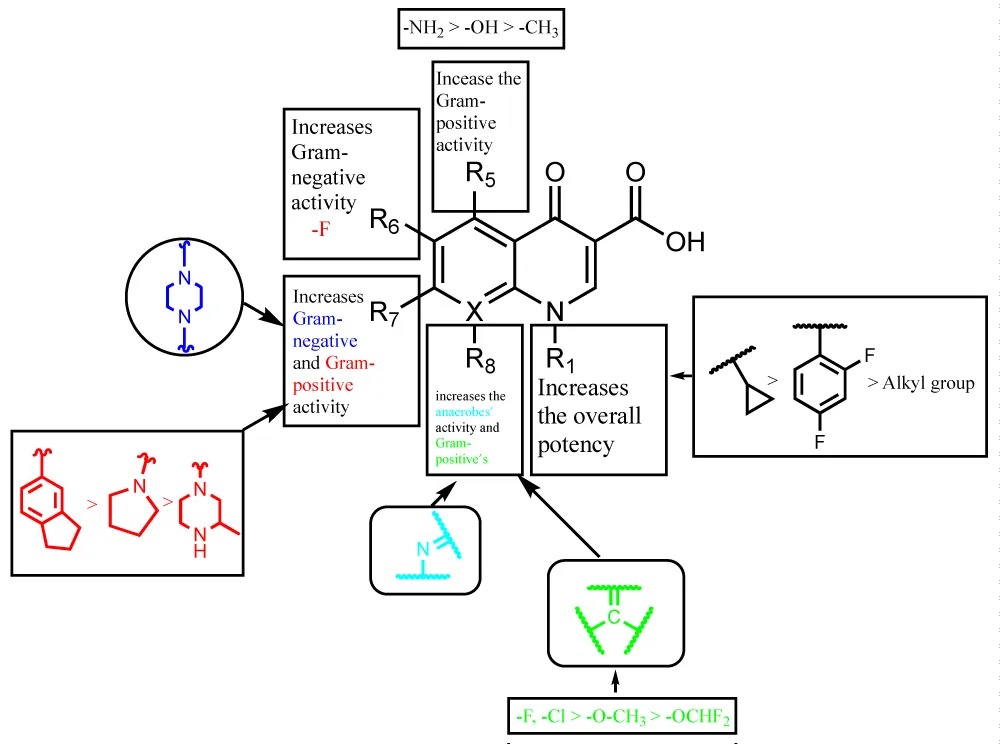
There are many FQs’ drug designs each serving an aimed purpose. For the inhibition of Gram-negative pathogens, Enoxacin, Norfloxacin, and Ciprofloxacin are the leading drugs as the addition of piperazine to the R7 position increased the activity against Gram-negative organisms. Whereas, when the modification was done on R1 by the addition of a cyclopropyl group, the overall potency of the drug was enhanced. Apart from the latter, extensive studies were made on the improvement of the activity of fluoroquinolones upon the structural modifications and all could be summarized in Figure 3. Figure 3 shows that a potent drug against Gram-positive and Gram-negative organisms could be designed as well as upon the aim, of different compounds could be synthesized [7].
Mechanisms
Depending upon the concentration of quinolones, their lethality towards bacteria varies. In recent years, the mechanism for each was thoroughly studied and many conclusions were drawn from the result. Quinolones are capable of killing bacteria either rapidly or slowly. When the concentration of quinolones is two-fold of the value of Minimum Inhibitory Concentration (MIC) the death of bacteria will be brought forward in an overnight period, whereas when the concentration of quinolones is five- to ten-fold of MIC, death of bacteria would happen in few hours. Slow death is well understood, yet the rapid is still under investigation [8].
Rapid death and slow death
In general, Quinolones’ bacterial lethality happens through the inhibition of the replication pathway of their DNA. DNA, during the stage of synthesis, opens up its double strands allowing the transcription with the complementary base pairing to occur. The enzymes responsible for such action in bacteria are known as topoisomerase-II, DNA gyrase, and DNA topoisomerase IV [7]. Once quinolones enter the scene, the inhibition could happen in two manners depending upon the concentration. Slow death is brought by blocking the replication and transcription of DNA gyrase and DNA topoisomerase IV. On the other hand, rapid death occurs when topoisomerase-II is removed and thus the two strands are unpaired which leads eventually to bacterial cell death through fragmentation of chromosomes. Nevertheless, since the whole process is reversible, any of these two approaches might end up in the accumulation of reactive oxygen species that would result in the quick death of the bacterial cell [8].
Mechanism of resistance
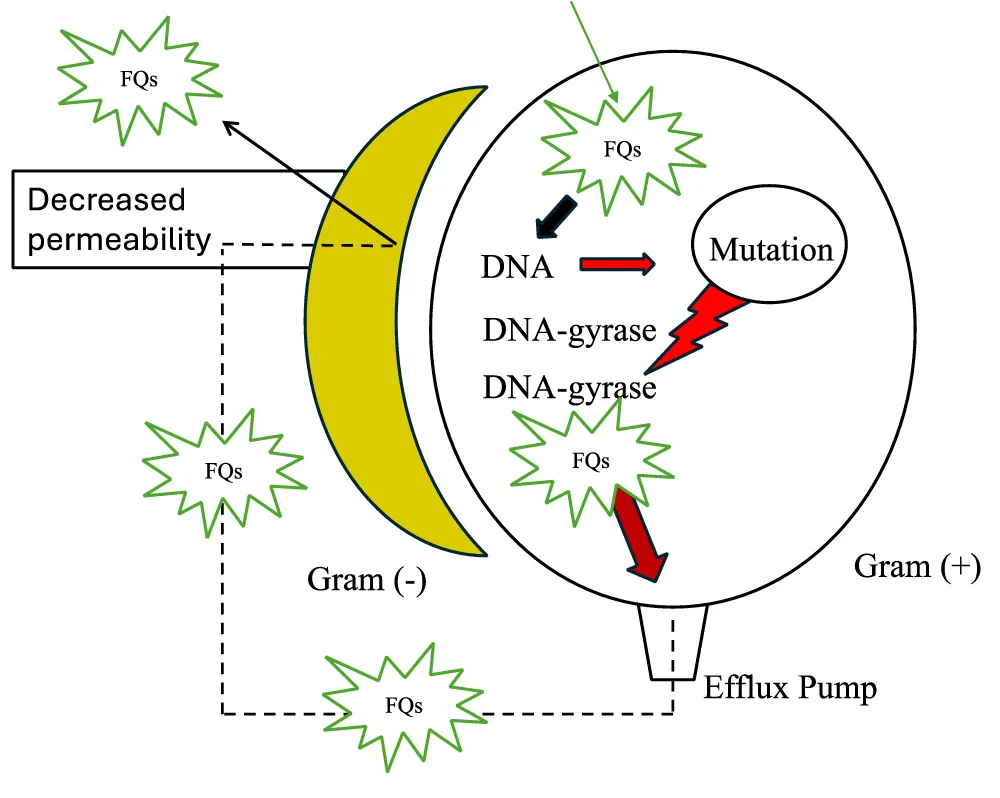
Although FQs are effective in their anti-bacterial activity, as a result of the mutation in targeted enzymes, namely DNA gyrase and DNA topoisomerase IV, or by inhibited accumulation of FQs as a result of efflux system their efficiency drops to the minimum leading to a resistance, Figure 4 [9]. Bacterial efflux pumps were first discovered during the 1980s specifically for tetracycline resistance in enterobacteria and with the passage of the years it was discovered that these pumps are capable of exerting all types of bacterial cytotoxic compounds including antibiotics. The excretion of antibiotics through these pumps could be either selective or non-selective. For the selective activity, as the name implies, the transportation of such substrates shall be confined to a certain type of relatively toxic substrates, whereas in the other case, pumps would be capable of transporting a wide variety of substrates such as dyes, disinfectants, antibacterial… etc. At physiological conditions, efflux pumps could increase the MIC from two up to eight folds thus contributing drastically to MDR [10]. The continual and frequent exposure of FQs by bacteria would lead eventually to resistance through alteration of gene expression which leads to hyperactivity of efflux pumps [9].
Occurrence and fate, risks and issues
FQs are widely used for the treatment of infectious diseases as they are of Gram-positive and Gram-negative activity and thus their consumption is very high. In 2009 it was estimated that worldwide they represented 17% of the sales in the antibiotics market, generating 7.1 billion US dollars, which indeed kept them in the third largest sales group in the pharmaceutical industry. As a consequence of the enormous consumption rate, a dreadful threat has emerged [11]. When FQs are metabolized, they are not fully metabolized: partially, which leads to once they are excreted in urine and feces to disaster as they are still in active form. These metabolites find their way to the environment as wastewater treatment plants are not capable of removing them, at least in the meantime [1].
Such toxic metabolites are generated generally from sewage effluents, animal waste discharge, and surface runoff. still, the largest amounts are released from residential areas, industries, and hospitals. Upon this discharge, FQs metabolites get spread across the environment; in recent studies, they have been found in soil, rivers, groundwater, and biological samples [11]. Leaching of FQs is a major concern as it would lead ultimately to MDR as well as contamination of soil, water, and food chains [1].
Risky it is indeed to keep blind eyes towards this issue, thus knowing the concentration of FQs and their metabolites in wastewater would be the first step towards removing them and retrieving the environment as sound as it was. In the coming section, detection and removal techniques during recent years shall be covered.
Detection and removal techniques
Up to this day, many attempts have been made towards the detection and removal of FQs from wastewater. The scientific community is marching ahead toward resolving this rather grueling issue.
Detection
In recent years, many analytical methods have been employed to detect FQs in wastewater such as gas chromatography-mass spectrometry (GC-MS), UV-visible spectroscopy, Liquid Chromatography-Mass Spectrometry (LC-MS), High-Pressure Liquid Chromatography (HPLC) [1], surface-enhanced Raman spectrometry, liquid chromatography-fluorescence detector (LC-FLD), voltammetry, chemiluminescence, colorimetry, and spectrofluorimetry [12]. Along with these analytical methods, a problem arises which is the difficulty in the removal of FQs from the matrix they dwell as they present in quite trace amounts. This issue compels the analyst to go through preconcentration steps such as normal liquid-liquid extraction, dispersive liquid-liquid extraction, and Dispersive Solid Phase Extraction (DSPE) [1]. Each of these has its limitations and thus covering the shortcomings is of great importance.
Attempting to investigate the presence of FQs in wastewater, Zhang, et al. [11] detected a total of 29 antibiotics in wastewater in China including FQs using HPLC-MS. Norfloxacin, Ciprofloxacin, Enrofloxacin, Ofloxacin, and Enoxacin were found with varying concentrations in influent and effluent wastewater. In influent and effluent wastewater, Ofloxacin was of the highest concentration ranging from 87.9 to 2032.3 ng/L and 1.2 to 498.8 ng/L, respectively. In the second place, Norfloxacin was of a concentration of 1.6 to 1454.5 ng/L in both wastewaters. This research was taken to another level; employing removal techniques such as activated sludge, and anoxic and oxic treatment to produce unharmful products. The efficiency of removing the latter FQs using such techniques ranged from 32.2% to 100%. In another study, cyclodextrin polymer capable of absorbing FQs in trace amounts as a preconcentration was synthesized before HPLC analysis. Cyclodextrin is a porous polymer that is used in the DSPE phase. In this research, fleroxacin, norfloxacin, ciprofloxacin, and gatifloxacin were analyzed. The Limit Of Detection (LOD) was 2.67, 3.17, 4.75, and 5.50 ng mL-1, respectively with a standard deviation of less than 4.5%. finally, the percentage of recovery was high in the range of 96.43% - 103.3% [13].
Detection of FQs is rather a grueling issue as they present in trace amounts and present in complex matrices therefore it is considered time-consuming to use the conventional ways of detection. Fluorescent Carbon Dots (CDs) provide a decent alternative to the latter as they are of low cost and high detection efficiency. Nitrogen Fluorescent Carbon Dots (N-CDs) were synthesized with a 20.5% quantum yield. These N-CDs are capable of emitting blue fluorescence radiation when excited at 368nm and quenched when Copper ion (Cu2+) is present. The fluorescence response is recovered upon the interaction with FQs especially Enrofloxacin which is an FQ. Thus, the detection is based on the recovery of fluorescence of N-CDs. When N-CDs were used a linear relationship was obtained from 0.1 to 15 µg mL-1 with LOD 0.16 µg mL-1 [14]. In another study, ciprofloxacin was the analyte using fluorescent CDs, the results showed that such CDs are of high selectivity and sensitivity towards ciprofloxacin with LOD 0.0002 µg mL−1when CDs were excited at 400 nm [15]. Korah, et al. have synthesized novel CDs by hydrothermal method starting from Curcuma amada. These CDs get excited at 360 nm and relax to give fluorescence at 445 nm. Not only were these CDs able to detect FQs rather they served duel-sensing purposes; sensing FQs as well as tetracyclines. The linear response was in the range of 2 nmol L-1 for FQs and LOD of 0.5 nmol L-1. Due to the enormous properties of CDs, they have attracted a great deal of attention from scientists to work them out as sensors. Bright yellow, fluorescent CDs (Y-CDs) were synthesized in one step through the hydrothermal process using o-phenylenediamine and 4-aminobutyric acid as precursors. Such CDs were capable of sensing FQs in the range of 17 - 67 nmol L-1 [16]. determination of norfloxacin and ciprofloxacin was achieved in combination with magnetic solid phase extraction using sulfur-doped carbon dots (S-CDs). S-CDs were synthesized from poly(4-styrenesulfonic acid-co-maleic acid) that are excited at 324 nm and relaxed back at 412 nm. The obtained LOD for ciprofloxacin was 6.7 nmol L-1 and for norfloxacin 4.6 nmol L-1. The linear range was 0.02 - 1.25 μmol L-1 and 0.02 - 1.0 μmol L-1 for ciprofloxacin and norfloxacin respectively [12].
Supramolecular assembly is one of the promising techniques for detection. In a research conducted by Shi, et al. [17] cucurbit[6]uril-based was synthesized starting with 4,4′-biphenyldisulphonic acid which showed high sensitivity through quenching of luminescence towards two FQs, namely levofloxacin, and gatifloxacin.
Removal
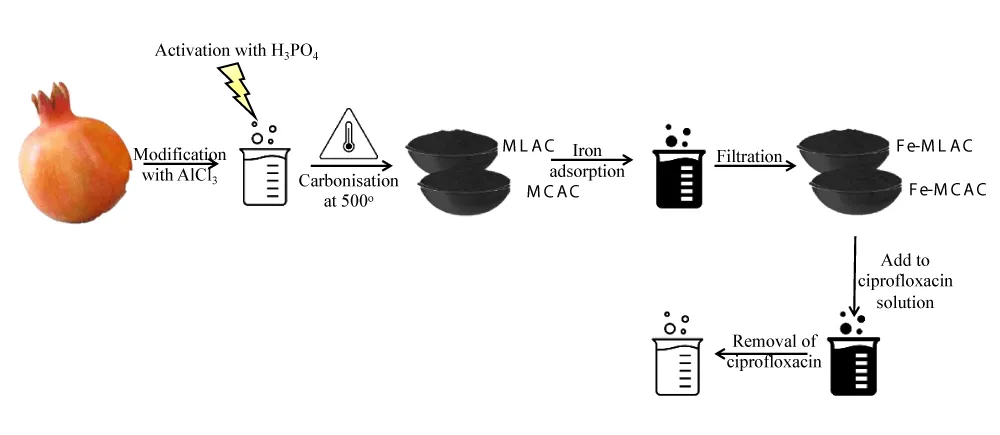
In recent years, the risk of keeping FQs running free through the ecosystem was assigned and upon such assignment, the removal of them was crucially important as many destructions in the balance of the environment could be lost as a result. Removal of FQs from wastewater was accomplished through the application of many different adsorbents in past years, such as montmorillonite, activated sludge-derived activated carbon, kaolinite, MOFs, modified coal fly ash, Titanium oxide aggregates, biochar … etc. each of which has its drawbacks and advantages [18]. The cost and efficiency of any adsorbent are of great concern, and they are crucial indeed to be under sight. The latter were forcing scientists to develop green and relatively cheap adsorbents to achieve the aim in mind. Annually, over 4 million tons of pomegranate are produced, and their peels represent 60% of their total weight. This grandiose amount could be used to synthesize rather efficient adsorbents for antibiotics such as ciprofloxacin. In a study, pomegranate peel’ nanostructured activated biochar was synthesized and its capacity to absorb ciprofloxacin from wastewater was studied. Upon the study, nanostructured activated biochar’s efficiency was determined to be 89.94% for batch reactor and 84.74% using packed-bed reactor. This rather outstanding capacity was attributed to the dominant interactions, namely: hydrogen bonding, pi-pi interaction, and electrostatic interactions [19]. The number of cycles that are to be done by an adsorbent is tremendously important, as it will -willy-nilly- reduce or increase the cost of the treatment regardless of the initial cost. Regeneration of the adsorbent is considered a cost reduction that takes the adsorbent in question to another level. Luffa sponges and corncobs a point time of their lives are considered as waste. This relative waste could be used to generate an outstanding adsorbent for FQs, especially ciprofloxacin which was done in a study conducted by El Bendary, et al. [18]. In this study, these residuals were used to produce activated carbon. Iron ions therein were adsorbed on the produced activated carbon through stirring in Fe(3+) solution. After filtration, the residuals were dried and thence used to remove ciprofloxacin Figure 5. Upon usage, the adsorption capacity was determined to be 476.19 and 416.67 mg g-1 for modified corn cobs activated carbon and modified luffa activated carbon, respectively with five recycles.
MXenes are a class of two-dimensional structures that are usually the result of selective etching of elements from 3A and 4A groups from the MAX phase that produces metal-nitrite/carbide. The safeness, high chemical durability, conductivity, and water affinity enabled MXenes to be highly applicable in environmental processes such as water treatment [20]. Recently, 2-D Ti3C2MXenes nanosheets attached to sodium ions were fabricated to remove ciprofloxacin from wastewater [21]. What is special regarding this research is the usage of these nanosheets as regenerative adsorbents through the passage of electrical current in a process called electrochemical regeneration. In electrochemical regeneration, the adsorbent is brought to its initial state through the electrochemical oxidation of the pollutants via electrical current [22]. The latter nanosheets were capable of removing ciprofloxacin by 99.7% from wastewater with stable results for 5 consecutive regenerated cycles [21]. Another useful class of crystals used for environmental remediation is metal-organic frameworks (MOFs) which are a unique microporous crystalline hybrid organic and inorganic material that have tuneable structural features that allow them to be used for absorption and separation technologies. Among the used MOFs are Zeolitic Imidazolate Frameworks (ZIF). ZIF-8 has been used to remove FQs such as ciprofloxacin, but in a recent study, it was used too to remove ofloxacin. The choice of using ZIF-8 to remove ofloxacin was mainly due to the presence of C-NH bonds in ZIF-8 and the presence of fluorine atom on ofloxacin that would allow the formation of hydrogen bondings as well as the presence of -COOH on ofloxacin that would coordinate with the unsaturated divalent metal during the self-assembly of ZIF-8. The results showed that ofloxacin was removed by ZIF-8 with maximum value of 96.3 ± 1.1% when it was introduced to 100mg L-1 solution of the latter. The availability for reusage was invest aged was well that showed upon four cycles the efficiency dropped just to 80.2% [23]. Wang, et al. [24] synthesized Cu-ZIF-8’s to remove ofloxacin from wastewater through impregnation of Cu2+ in ZIF-8. The latter adsorbents were capable of removing ofloxacin 4.2 times more than ZIF-8 alone. Unlike ZIF-8, Cu- ZIF-8 could be generated easily upon washing with ethanol. Besides ZIFs, Materials of Institute Lavoisier frameworks (MIL) are highly applicable as they are of high stability, wide porosity, and large surface areas that make them ideal for water treatment as adsorbents [25]. A novel MIL of 53(Fe-Cu) was synthesized through a solvothermal single-step route. The previous MILs were used in treating quite a lot of pharmaceuticals in wastewater including ciprofloxacin. For ciprofloxacin, the maximum adsorption capacity was obtained to be 190.4 mg g-1, with a maximum degradation efficiency of 74.48% while using UV light and 57.88% when irradiation with visible radiation [26].
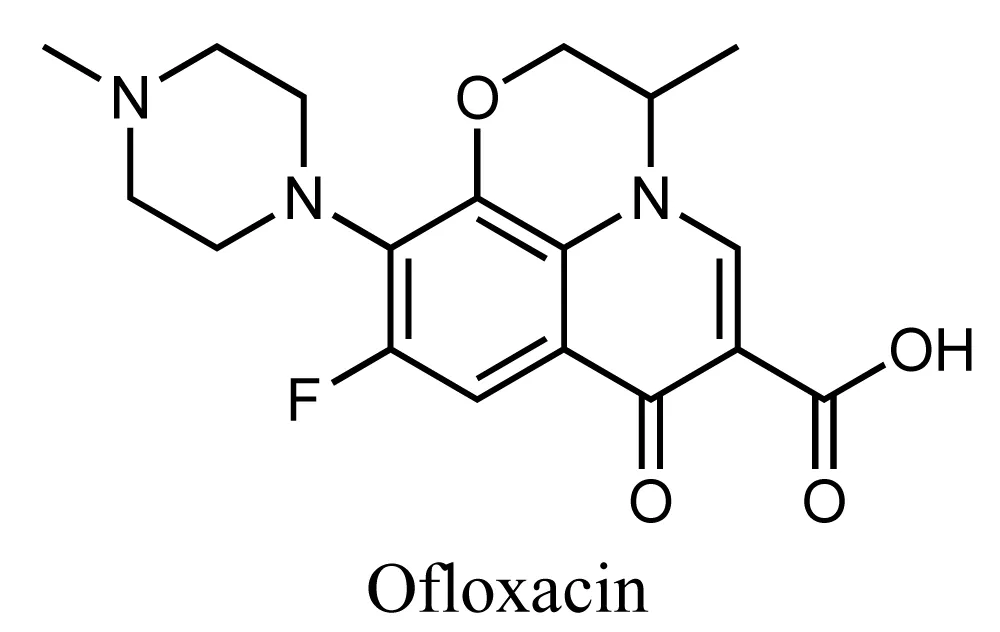
Novelty does not have a limit and new methods are invented daily. A combination of TiO2 Graphitic Carbon Nitride (g-C3N4) on activated carbon was prepared recently through the gel method. The latter can degrade levofloxacin via photoelectric activity in wastewater. The previous conclusion was drawn as the electrode’s degrading capability was studied under different conditions and it was found that the most suitable conditions as 94.76% was degraded in 4 hours were when the pH is 3 with an aeration rate of 8L min-1, 8% mass ratio between g-C3N4 and TiO2, respectively and with ultrasonic 200W. the electrode is of great potential as after 6 recycles, the degradation level was 71.17%. which is still enormous indeed. The degraded product was antibiotically inactive, and they were analyzed on different bacterial colonies as well as analyzed using HPLC-MS [27]. Ofloxacin is one of the FQs that inhibits the replication and synthesis of bacteria’s DNA. Each FQ has its functional groups that its bioactivity is attributed to. The presence of two core rings which include pyridine and benzene rings that are substituted with fluorine is the main suggested reason for the activity of Ofloxacin, Figure 6.
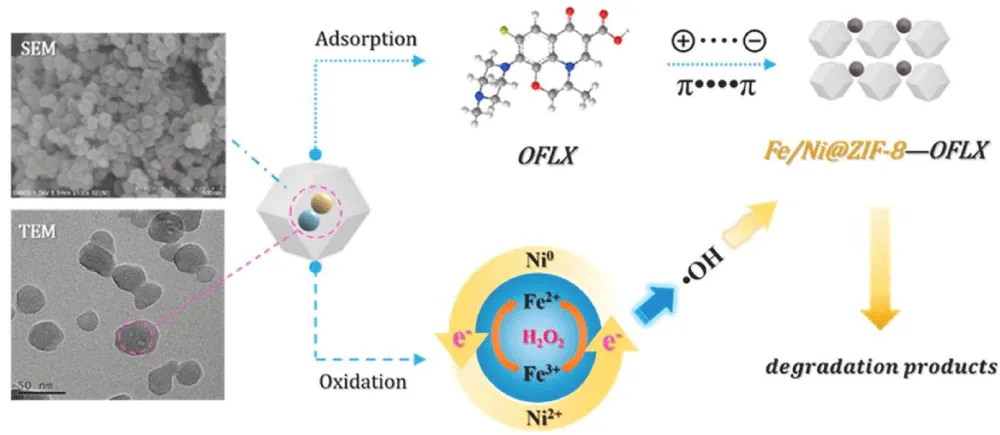
The attention towards Carbon Nitride (C3N4) increased in recent years as it has an outstanding response towards light and is a green chemical as well, yet it has a shortcoming: the photo-induced electrons recombine with C3N4’s holes rapidly, which limits its applications. The latter shortcoming was diluted by Shang, et al. [28] as a photocatalyst composite of titanium silicon molecular sieve loaded C3N4 (TS-1/C3N4) was synthesized and used to photodegrade Ofloxacin. The study investigated the possible conditions, and 82.92% removal efficiency was obtained under: 1.55 g L-1 of catalyst, loading of 58.60% of TS-1, and luminous power density of 49.38 mW cm-2. In a study held in 2022, Fe/Ni@ZIF-8 composite was synthesized and used to remove and degrade Ofloxacin through Fenton-like oxidation. Fenton oxidation is the process that uses H2O2 that was activated by Iron salts, which could be summarised as follows, Equation (1) [29].
Equation 1: Overall Fenton Equation
The previous composite was able to remove Ofloxacin with an efficiency of more than 98% under 45oC, pH = 6, 10mM of H2O2, [Ofloxacin]i = 10 mg L-1, with maximum adsorption of 154.55 mg g-1. The stability of the synthesized composite was reflected in the number of cycles (five cycles) in which it could be used and still having good adsorbability. Real wastewater samples were used as well and the removal efficiency was above 90% [30]. The whole study is summarised in Figure 7.
Homogeneous Fenton reactions operate under a limited range of pH as well as the recovery of iron pollutants is rather grueling which indeed is to be overcome by using a heterogeneous system. Heterogeneous Fenton reactions involve the usage of Fe3O4 nanoparticles and a source of radiations which help in the reduction of iron ions concentrations and therefore allow functioning in a wider range of pH. Pulsed Discharge Plasma (PDP) is a non-thermal plasma that is used to decontaminate aqueous pollutants through oxidation. Such a method provides rapidity, efficiency, and production of no secondary pollutants. Graphene-Fe3O4 was coupled with PDP to degrade Ofloxacin in wastewater. The nanocomposites first were synthesized through the hydrothermal method, comparing the degradation capacity of PDP alone and the coupled PDP nanocomposite it was reported that the efficiency increased from 65% to 99.9%. the oxidized chemicals were also analyzed using LC-MS, and IC their toxicity was reduced enormously [31].
Advanced oxidation processes (AOPs) have received tremendous attention in the last decade in the field of antibiotics degradation as they are of rapidity, and high capacity for oxidation of antibiotics in aqueous media [32]. Out of the used AOPs, UV-H2O2 is widely applicable due to its high capacity in water treatment against antibiotics and its relatively low cost. Nie, et al. [33] used the latter method to degrade Ofloxacin in wastewater through the effect of alumina/silica-based oxide-suspended particles. The results showed that silica oxide suspended particles have no effect on the degradation capacity whereas it was enhanced upon the usage of alumina oxide. Under alumina, a pH range of 3 - 5 was giving the best removal results. The latter performance was explained through the ability of suspended particles to provide a large surface area and thus promote the production of •OH. Another recently developed method for the degradation of antibiotics in wastewater is a sono-Fenton reaction. Sono-Fenton is a combination of ultrasound and Fenton mechanisms that would reduce the time required for oxidation as well as make the oxidation more selective. The reaction starts with sonolysis which is the formation of cavitations that will eventually collapse and emit high pressure and temperature. Such high energy is capable of cleaving molecules through pyrolysis, including water, and forming radicals. Equation (2) illustrates the latter [34].
Pollutants+))) → Degraded pollutant
H2O + • OH + • H
Pollutants + • H → Degraded pollutant
))) = Ultrasound radiations
The previous method was used recently to degrade Ciprofloxacin in wastewater by González, et al. [35] as they reported that the best degradation rate was obtained when 580 kHz frequency was used during 15 minutes at 14.2mM of H2O2 and H2O2/Fe2+ = 6 that degraded 60% of the available Ciprofloxacin.
The fusion of natural minerals to enhance the environmental situation is of outstanding benefit as these minerals are considered low cost and high abundance. Ilmenite, which is a natural mineral consisting of Fe2-xTixO3 is highly abundant and is known to be of high biocompatibility and friendliness to the environment. Recent studies showed that Ilmenite can bind to hydroxyls and form active sites which allow their usage as adsorbents to environmental pollutants. Lui, et al. [36] prepared an ilmenite biochar composite through the co-cracking of corn stover and natural ilmenite so that it could be used to remove ofloxacin from wastewater. 90% removal capacity was obtained in the pH range of 2-10 which was the result of hydrogen bonding (-COOH and -OH), π-π interactions, and pore filling mechanism.
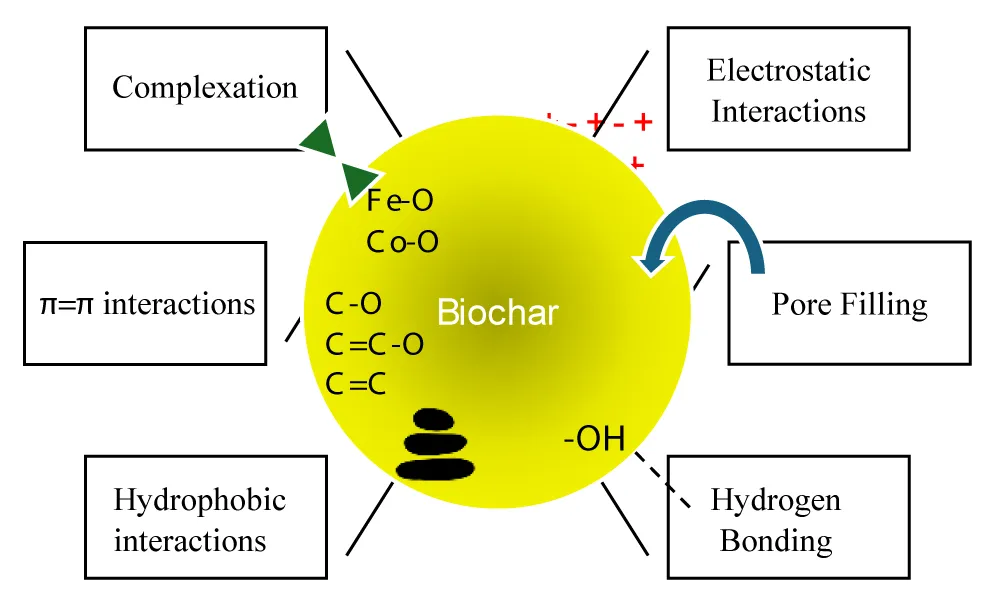
The pyrolysis of forestry waste cedar bark is a source of biochar that was used by Hao, et al. [37] to synthesize Fe-Co-modified biochar. For the latter biochar, the best adsorption capacity was at pH =7. And after five cycles the capacity was between 75.78% - 89.31%. Figure 8 shows the different mechanisms by which biochar is capable of adsorbing Ofloxacin.
Bismuth tungstate Bi2WO6 has good photocatalytic properties under the range of visible radiation, yet not the best [38]. To enhance its photocatalytic activity, Wang, et al. [39] coupled the latter with biochar and Fe3O4. During the study, it was reported that under 30 minutes frame of time, complete degradation of ofloxacin and ciprofloxacin happened which was attributed to the production of radical •O2−.
Nanoparticles are other adsorbents that are suggested to be of great use to remove pharmaceuticals from wastewater. Among the recent nanoparticles used are Zinc Oxide and graphene oxide nanoparticles (ZnONPs, GONPs), the latter were used to remove Levofloxacin from wastewater. Levofloxacin is another FQ that can seep into the aquatic system and cause severe threats such as antibiotic-resistant bacteria and mutagenicity. The synthesized nanoparticles were used as adsorbents as well as photocatalysts to degrade the adsorbed antibiotic. The study was done against various parameters such as pH, UV radiation, the concentration -of antibiotics and nanoparticles- etc. the main findings were that the highest removal rate was when both nanoparticles, namely: ZnONPs/GONPs, were used which gives a chance for the researchers to go through deep study of the optimum conditions needed to remove other FQs from real samples [40]. Moringa Olifera contains a significant quantity of polyphenols, terpenoids, and flavonoids which act as stabilizing and capping agents during the synthesis of nanoparticles. The latter was used to synthesize magnetite nanoparticles through green synthesis, namely F3O4 - gINPs. After synthesis and characterization, the previous nanoparticles were used to remove Levofloxacin from wastewater. The mechanism of removal was investigated, and it was suggested from the data that it is chemisorption. The capacity of removal was outstanding as it was close to 86.15% with a maximum absorption of 22.47 mg g-1 [41].
Photodegradation is significant, yet it has its drawbacks, such as the toxicity of the catalyst used which indeed is a grueling matter. Thus, green methods are much more preferable. Recently, Sun & Zheng, 2022 [42], used a rather green method to eliminate the presence of such pollutants in the environment: biodegradation. Biodegradation is the most useful among the other methods in the removal of FQs, the author says; as the products out of such method shall be not biotoxic, not polluting to the environment, not corrosive, and of long effectiveness. Along with the latter, it requires low capital. In their research, wastewater was allowed to run across a Vertical Flow Constructed Wetland (VFCW) which consists of two main parts. The first part was a substrate layer that is capable of removing pollutants through the presence of pollution-resistant, flood-resistant, and plants full of roots, and the other was a layer to support the system. In this method, wastewater containing FQs is introduced to the system and FQs are trapped as a result by plants. Once they are trapped, they undergo a biodegradation process either aerobic or anaerobic. In this study, the flow rate across the VFCW was varied and it was suggested that in both cases -high and low flow rates- the efficiency of removal was high: > 95% and 80% respectively. Finally, it was suggested too that the microorganisms responsible for such biodegradation are Flavobacterium, Microbacterium, and Dechloromonas. It was found too, using the same removal technique that Devosia, Pseudorhodoferax, Cellvibrio, Bosea, Caulobacter, Acinetobacter, Zoogloea, Arcobacter, Dechloromonas, Flavobacterium, Nakamurella, Chloroplast, Clostridiumsensustricto1, Pelosinus, UTCFX1 and Hypnocyclicus microorganisms are essential to removal of FQs from VFCW and the high efficiency of such method is attributed to their presence [43]. In another method, activated carbon-decorated polyacrylonitrile nanofibers were employed as adsorbents for selected FQs, namely ciprofloxacin, danofloxacin, and enrofloxacin. The synthesized nanofibers were able to adsorb FQs with LOD of 0.05, 0.11, 0.20, and 0.53, 1.21, 2.17 mg L-1 [1].
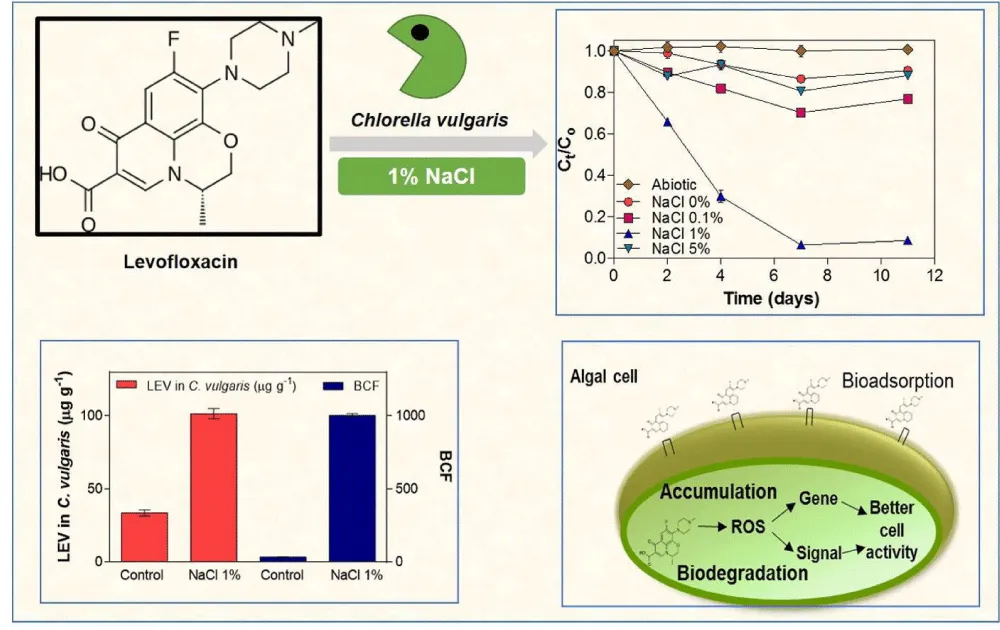
FQs are considered as Endocrine-Disrupting Chemicals (EDCs). EDCs are exogenous chemicals that alter the function of the endocrine systems thus leading to severe health problems in an organism or its progeny [44]. During the last couple of years, many methods were proposed to degrade ECDs including FQs. Among the latter, Chlorella vulgaris (C. vulgaris) was considered among the pioneers as it has a high capacity to absorb and degrade the typical EDCs in an aqueous medium [45]. In research, six different microalgae, namely: Chlamydomonas mexicana, Chlamydomonas pitschmannii, C. vulgaris, Ourococcus multisports, Micractinium dresser, Tribonema adequate were used to degrade levofloxacin in water in which its presence in the aqua sphere might lead to development of bacterial resistant through the delivery of drug to non-targeted species. The results showed that C. vulgaris was of the highest degradation rate as in 11 days more than 80% of levofloxacin was degraded. The previous results were attributed to C. vulgaris’s’ capability to bioaccumulate and then subsequently degrade intracellularly through the salinization of water Figure 9 [45]. Researchers were held then upon these microalgae to behold where their limitations lay. Wu, et al. [46] studied the latter and concluded that the presence of microplastics such as Polystyrene microplastics inhibits the growth of C. vulgaris and thus their potential application as removal agents of FQs would be in vain. From the previous research it is worth attention that upon the presence of microplastics in water, this method would be non-functional, and therefore, previous determination of the content of water composition against microplastics would be truly helpful before going ahead using this treatment method.
The usage of electrochemical techniques to remediate wastewater from antibiotics is of great importance. Among the recently developed techniques is Electrocoagulation (EC). EC is an electrochemical process that generates coagulant species by oxidation of materials that stick upon the anode selectively by the application of a specific current. Iron and aluminum are the most applied electrodes in this technique due to their wide availability, non-toxicity, and high valence that allows for the efficient removal of various pollutants [47]. Up to 2017, there was no application of EC to remove FQs from wastewater, Ahmadzadeh, et al. [48] were the leaders in this application. During the study, the conditions required to obtain the best efficiency were investigated and it was reported to be at pH 7.78 with 1cm as an inter-electrode distance, allowing 20 minutes to be reaction time, density of current 12.5 mA.cm-2 and dose of electrolyte of 0.07 M NaCl with the initial concentration of Ciprofloxacin 32.5 mg L-1 to obtain 88.57% removal efficiency. In 2022, EC was applied for the first time to remove ofloxacin from wastewater. The aluminum electrode was successfully capable of removing ofloxacin by 70% within 40 minutes and the maximum removal capacity was reached in 105 minutes which was 78%. Such efficiency was explained by the surface complexation that involves the functional groups located at the electrode’s surface namely, triple bond aluminium-OH and the deprotonation and protonation of ofloxacin (ofloxacin- and ofloxacin’s zwitterion) [49]. The application of EC in the removal of FQs was developed since its first application and recently, hydrogen was produced out of the degradation of FQs using EC coupled with photocatalysis. A complete reactor was designed by Muttaqin, et al. [50] in which the anode was acrylic equipped with aluminum and the cathode was plates of 316 stainless steel. The used photocatalyst was Fe-doped titania nanotube arrays. Using a 250-W mercury lamp as the light source, the degradation process took place, and hydrogen gas was produced as well as a result of H+ reduction on the cathode.
Conclusion and recommendations
In conclusion, FQs are a grave threat to the ecosystem that must be dealt with properly. In recent years, scientists recognized the latter issue and thus they drove new methods to circumvent the problem. Different methods and techniques were developed during the past few years, each of which has its advantages and disadvantages. The detection of FQs was best upon using CDs as they are of low cost and efficient detection through their fluorescent recovery when interacting with FQs. The removal of FQs is of great benefit to the environment as a whole, 2-D Ti3C2MXenes nanosheets attached to sodium ions achieved the best removal rate which is 99.7% for ciprofloxacin, yet the other listed materials were of varying efficiencies. recommended to have advanced wastewater treatment plants that are using the methods highlighted priori, yet industrial scale techniques are not reported which could be considered as a shortcoming in the literature, and thus it is recommended as well to study the behavior of the methods mentioned above to have real-applications in the future.
- Mogolodi Dimpe K, Nomngongo PN. Application of activated carbon-decorated polyacrylonitrile nanofibers as an adsorbent in dispersive solid-phase extraction of fluoroquinolones from wastewater. J Pharm Anal. 2019 Apr;9(2):117-126. doi: 10.1016/j.jpha.2019.01.003. Epub 2019 Jan 10. PMID: 31011468; PMCID: PMC6460376.
- Petrie B, Barden R, Kasprzyk-Hordern B. A review on emerging contaminants in wastewaters and the environment: current knowledge, understudied areas and recommendations for future monitoring. Water Res. 2015 Apr 1; 72:3-27. doi: 10.1016/j.watres.2014.08.053. Epub 2014 Sep 10. PMID: 25267363.
- Tanwar J, Das S, Fatima Z, Hameed S. Multidrug resistance: an emerging crisis. Interdiscip Perspect Infect Dis. 2014; 2014:541340. doi: 10.1155/2014/541340. Epub 2014 Jul 16. PMID: 25140175; PMCID: PMC4124702.
- Leng L, Wei L, Xiong Q, Xu S, Li W, Lv S, Lu Q, Wan L, Wen Z, Zhou W. Use of microalgae based technology for the removal of antibiotics from wastewater: A review. Chemosphere. 2020 Jan; 238:124680. doi: 10.1016/j.chemosphere.2019.124680. Epub 2019 Aug 26. PMID: 31545213.
- Khan NA, Ahmed S, Farooqi IH, Ali I, Vambol V, Changani F, Yousefi M, Vambol S, Khan SU, Khan AH. Occurrence, sources and conventional treatment techniques for various antibiotics present in hospital wastewaters: A critical review. TrAC - Trends in Analytical Chemistry. 2020; 129. https://doi.org/10.1016/j.trac.2020.115921
- Coates AR, Halls G, Hu Y. Novel classes of antibiotics or more of the same? Br J Pharmacol. 2011 May;163(1):184-94. doi: 10.1111/j.1476-5381.2011.01250.x. PMID: 21323894; PMCID: PMC3085877.
- Pham TDM, Ziora ZM, Blaskovich MAT. Quinolone antibiotics. Medchemcomm. 2019 Jun 28;10(10):1719-1739. doi: 10.1039/c9md00120d. PMID: 31803393; PMCID: PMC6836748.
- Bush NG, Diez-Santos I, Abbott LR, Maxwell A. Quinolones: Mechanism, Lethality and Their Contributions to Antibiotic Resistance. Molecules. 2020 Dec 1;25(23):5662. doi: 10.3390/molecules25235662. PMID: 33271787; PMCID: PMC7730664.
- Brar RK, Jyoti U, Patil RK, Patil HC. Fluoroquinolone antibiotics: An overview. Adesh University Journal of Medical Sciences & Research. 2020; 2(1):26–30. https://doi.org/10.25259/aujmsr_12_2020
- Zechini B, Versace I. Inhibitors of multidrug resistant efflux systems in bacteria. Recent Pat Antiinfect Drug Discov. 2009 Jan;4(1):37-50. doi: 10.2174/157489109787236256. PMID: 19149695.
- Zhang X, Zhao H, Du J, Qu Y, Shen C, Tan F, Chen J, Quan X. Occurrence, removal, and risk assessment of antibiotics in 12 wastewater treatment plants from Dalian, China. Environ Sci Pollut Res Int. 2017 Jul;24(19):16478-16487. doi: 10.1007/s11356-017-9296-7. Epub 2017 May 28. PMID: 28551746.
- Hua J, Jiao Y, Wang M, Yang Y. Determination of norfloxacin or ciprofloxacin by carbon dots fluorescence enhancement using magnetic nanoparticles as adsorbent. Mikrochim Acta. 2018 Jan 27;185(2):137. doi: 10.1007/s00604-018-2685-x. PMID: 29594473.
- Zhang J, Liu D, Shi Y, Sun C, Niu M, Wang R, Hu F, Xiao D, He H. Determination of quinolones in wastewater by porous β-cyclodextrin polymer based solid-phase extraction coupled with HPLC. J Chromatogr B Analyt Technol Biomed Life Sci. 2017 Nov 15;1068-1069:24-32. doi: 10.1016/j.jchromb.2017.09.046. Epub 2017 Oct 2. PMID: 29028615.
- Guo X, Zhang L, Wang Z, Sun Y, Liu Q, Dong W, Hao A. Fluorescent carbon dots based sensing system for detection of enrofloxacin in water solutions. Spectrochim Acta A Mol Biomol Spectrosc. 2019 Aug 5;219:15-22. doi: 10.1016/j.saa.2019.02.017. Epub 2019 Feb 11. PMID: 31030043.
- Gao X, Sun M, Liu X, Li X, Li J. A ratiometric fluorescence platform based on carbon dots for visual and rapid detection of copper(II) and fluoroquinolones. Mikrochim Acta. 2022 Mar 15;189(4):144. doi: 10.1007/s00604-022-05243-3. PMID: 35292904.
- Lu W, Jiao Y, Gao Y, Qiao J, Mozneb M, Shuang S, Dong C, Li CZ. Bright Yellow Fluorescent Carbon Dots as a Multifunctional Sensing Platform for the Label-Free Detection of Fluoroquinolones and Histidine. ACS Appl Mater Interfaces. 2018 Dec 12;10(49):42915-42924. doi: 10.1021/acsami.8b16710. Epub 2018 Nov 27. PMID: 30412373.
- Shi L, Liu M, Li H. A cucurbit[6]uril-based supramolecular assembly as a highly sensitive and quickly responsive luminescent sensor for the detection of fluoroquinolone antibiotics in simulated wastewater. CrystEngComm. 2020; 22(22):3753–3758. https://doi.org/10.1039/d0ce00603c
- El-Bendary N, El-Etriby HK, Mahanna H. Reuse of adsorption residuals for enhancing removal of ciprofloxacin from wastewater. Environ Technol. 2021 Jul 26:1-17. doi: 10.1080/09593330.2021.1952310. Epub ahead of print. PMID: 34223807.
- Hamadeen HM, Elkhatib EA. New nanostructured activated biochar for effective removal of antibiotic ciprofloxacin from wastewater: Adsorption dynamics and mechanisms. Environ Res. 2022 Jul;210:112929. doi: 10.1016/j.envres.2022.112929. Epub 2022 Feb 12. PMID: 35167852.
- Wei Z, Peigen Z, Wubian T, Xia Q, Yamei Z, ZhengMing S. Alkali-treated Ti3C2Tx MXenes and their dye adsorption performance. Materials Chemistry and Physics. 2018; 206:270–276. https://doi.org/10.1016/j.matchemphys.2017.12.034
- Ghani AA, Shahzad A, Moztahida M, Tahir K, Jeon H, Kim B, Lee DS. Adsorption and electrochemical regeneration of intercalated Ti3C2Tx MXene for the removal of ciprofloxacin from wastewater. Chemical Engineering Journal. 2021; 421(P2):127780. https://doi.org/10.1016/j.cej.2020.127780
- Narbaitz RM, Karimi-Jashni A. Electrochemical regeneration of granular activated carbons loaded with phenol and natural organic matter. Environ Technol. 2009 Jan;30(1):27-36. doi: 10.1080/09593330802422803. PMID: 19213463.
- Yu R, Wu Z. High adsorption for ofloxacin and reusability by the use of ZIF-8 for wastewater treatment. Microporous and Mesoporous Materials, 308(March). 2020; 110494. https://doi.org/10.1016/j.micromeso.2020.110494
- Wang X, Zhao Y, Sun Y, Liu D. Highly Effective Removal of Ofloxacin from Water with Copper-Doped ZIF-8. Molecules. 2022 Jul 5;27(13):4312. doi: 10.3390/molecules27134312. PMID: 35807556; PMCID: PMC9268637.
- Zhang H, Hu X, Li T, Zhang Y, Xu H, Sun Y, Gu X, Gu C, Luo J, Gao B. MIL series of metal organic frameworks (MOFs) as novel adsorbents for heavy metals in water: A review. J Hazard Mater. 2022 May 5;429:128271. doi: 10.1016/j.jhazmat.2022.128271. Epub 2022 Jan 14. PMID: 35093745.
- Chatterjee A, Jana AK, Basu JK. A binary MOF of iron and copper for treating ciprofloxacin-contaminated wastewater by an integrated technique of adsorption and photocatalytic degradation. New Journal of Chemistry. 2021; 45(37):17196–17210. https://doi.org/10.1039/d1nj02880d
- Xing Z, Wang Z, Chen W, Zhang M, Fu X, Gao Y. Degradation of levofloxacin in wastewater by photoelectric and ultrasonic synergy with TiO2/g-C 3 N 4 @AC combined electrode. J Environ Manage. 2023 Mar 15;330:117168. doi: 10.1016/j.jenvman.2022.117168. Epub 2023 Jan 3. PMID: 36603258.
- Shang Q, Liu X, Zhang M, Zhang P, Ling Y, Cui G, Liu W, Shi X, Yue J, Tang B. Photocatalytic degradation of ofloxacin antibiotic wastewater using TS-1/C 3 N 4 composite photocatalyst: Reaction performance optimization and estimation of wastewater component synergistic effect by artificial neural network and genetic algorithm. Chemical Engineering Journal. 2022; 443. https://doi.org/doi.org/10.1016/j.cej.2022.136354.
- Aramyan SM. Advances in Fenton and Fenton Based Oxidation Processes for Industrial Effluent Contaminants Control-A Review. International Journal of Environmental Sciences & Natural Resources. 2017; 2(4). https://doi.org/10.19080/ijesnr.2017.02.555594
- Zhang T, Wu P, Owens G, Chen Z. Adsorption and fenton-like oxidation of ofloxacin in wastewater using hybrid MOF bimetallic Fe/Ni nanoparticles. Chemosphere. 2022 Nov;307(Pt 2):135936. doi: 10.1016/j.chemosphere.2022.135936. Epub 2022 Aug 5. PMID: 35934098.
- Guo H, Li Z, Lin S, Li D, Jiang N, Wang H, Han J, Li J. Multi-catalysis induced by pulsed discharge plasma coupled with graphene-Fe 3 O 4 nanocomposites for efficient removal of ofloxacin in water: Mechanism, degradation pathway and potential toxicity. Chemosphere. 2021 Feb;265:129089. doi: 10.1016/j.chemosphere.2020.129089. Epub 2020 Nov 23. PMID: 33261841.
- Wang J, Zhuan R. Degradation of antibiotics by advanced oxidation processes: An overview. Sci Total Environ. 2020 Jan 20;701:135023. doi: 10.1016/j.scitotenv.2019.135023. Epub 2019 Nov 3. PMID: 31715480.
- Nie Y, Sun X, Wang M, Tian X, Yang C, Dai C. Natural alumina/silica suspended particles in water to enhance ofloxacin degradation with UVA-H 2 O 2 driven by surface chemistry. J Hazard Mater. 2021 Jun 15;412:125259. doi: 10.1016/j.jhazmat.2021.125259. Epub 2021 Jan 29. PMID: 33545644.
- Adityosulindro S, Barthe L, González-Labrada K, Jáuregui Haza UJ, Delmas H, Julcour C. Sonolysis and sono-Fenton oxidation for removal of ibuprofen in (waste)water. Ultrason Sonochem. 2017 Nov;39:889-896. doi: 10.1016/j.ultsonch.2017.06.008. Epub 2017 Jun 16. PMID: 28733020.
- González Labrada K, Alcorta Cuello DR, Saborit Sánchez I, García Batle M, Manero MH, Barthe L, Jáuregui-Haza UJ. Optimization of ciprofloxacin degradation in wastewater by homogeneous sono-Fenton process at high frequency. J Environ Sci Health A Tox Hazard Subst Environ Eng. 2018;53(13):1139-1148. doi: 10.1080/10934529.2018.1530177. Epub 2019 Jan 9. PMID: 30623707.
- Liu Y, Yuan Y, Wang Z, Wen Y, Liu L, Wang T, Xie X. Removal of ofloxacin from water by natural ilmenite-biochar composite: A study on the synergistic adsorption mechanism of multiple effects. Bioresour Technol. 2022 Nov;363:127938. doi: 10.1016/j.biortech.2022.127938. Epub 2022 Sep 12. PMID: 36100186.
- Hao J, Wu L, Lu X, Zeng Y, Jia B, Luo T, He S, Liang L. A stable Fe/Co bimetallic modified biochar for ofloxacin removal from water: adsorption behavior and mechanisms. RSC Adv. 2022 Nov 4;12(49):31650-31662. doi: 10.1039/d2ra05334a. PMID: 36380923; PMCID: PMC9634719.
- Saha D, Bøjesen ED, Mamakhel AH, Iversen BB. Why Does Bi2WO6 Visible-Light Photocatalyst Always Form as Nanoplatelets? Inorg Chem. 2020 Jul 6;59(13):9364-9373. doi: 10.1021/acs.inorgchem.0c01249. Epub 2020 Jun 22. PMID: 32567841.
- Wang Z, Cai X, Xie X, Li S, Zhang X, Wang Z. Visible-LED-light-driven photocatalytic degradation of ofloxacin and ciprofloxacin by magnetic biochar modified flower-like Bi2WO6: The synergistic effects, mechanism insights and degradation pathways. Sci Total Environ. 2021 Apr 10;764:142879. doi: 10.1016/j.scitotenv.2020.142879. Epub 2020 Oct 14. PMID: 33129540.
- El-Maraghy CM, El-Borady OM, El-Naem OA. Effective Removal of Levofloxacin from Pharmaceutical Wastewater Using Synthesized Zinc Oxid, Graphen Oxid Nanoparticles Compared with their Combination. Sci Rep. 2020 Apr 3;10(1):5914. doi: 10.1038/s41598-020-61742-4. PMID: 32246063; PMCID: PMC7125086.
- Altaf S, Zafar R, Zaman WQ, Ahmad S, Yaqoob K, Syed A, Khan AJ, Bilal M, Arshad M. Removal of levofloxacin from aqueous solution by green synthesized magnetite (Fe 3 O 4 ) nanoparticles using Moringa Olifera : Kinetics and reaction mechanism analysis. Ecotoxicol Environ Saf. 2021 Dec 15;226:112826. doi: 10.1016/j.ecoenv.2021.112826. Epub 2021 Sep 27. PMID: 34592521.
- Sun W, Zheng Z. Research on removal of fluoroquinolones in rural domestic wastewater by vertical flow constructed wetlands under different hydraulic loads. Chemosphere. 2022 Sep;303(Pt 3):135100. doi: 10.1016/j.chemosphere.2022.135100. Epub 2022 May 26. PMID: 35644233.
- Sun W, Zheng Z. Research on removal of fluoroquinolones in rural domestic wastewater by vertical flow constructed wetlands under different hydraulic loads. Chemosphere. 2022 Sep;303(Pt 3):135100. doi: 10.1016/j.chemosphere.2022.135100. Epub 2022 May 26. PMID: 35644233.
- Kürzinger L, Fassnacht M, Dischinger U. Endokrine Disruptoren [Endocrine Disrupting Chemicals]. Dtsch Med Wochenschr. 2022 Nov;147(22):1437-1443. German. doi: 10.1055/a-1837-2670. Epub 2022 Nov 1. PMID: 36318905.
- Xiong JQ, Kurade MB, Jeon BH. Biodegradation of levofloxacin by an acclimated freshwater microalga, Chlorella vulgaris. Chemical Engineering Journal. 2017; 313:1251–1257. https://doi.org/10.1016/j.cej.2016.11.017
- Wu X, Wu H, Zhang A, Sekou K, Li Z, Ye J. Influence of polystyrene microplastics on levofloxacin removal by microalgae from freshwater aquaculture wastewater. J Environ Manage. 2022 Jan 1; 301:113865. doi: 10.1016/j.jenvman.2021.113865. Epub 2021 Sep 29. PMID: 34597951.
- Shahedi A, Darban AK, Taghipour F, Jamshidi-Zanjani A. A review on industrial wastewater treatment via electrocoagulation processes. Current Opinion in Electrochemistry. 2020; 22:154–169. https://doi.org/10.1016/j.coelec.2020.05.009
- Ahmadzadeh S, Asadipour A, Pournamdari M, Behnam B, Rahimi HR, Dolatabadi M. Removal of ciprofloxacin from hospital wastewater using electrocoagulation technique by aluminum electrode: Optimization and modelling through response surface methodology. Process Safety and Environmental Protection. 2017; 109:538–547. https://doi.org/10.1016/j.psep.2017.04.026
- Mokni S, Tlili M, Jedidi N, Hassen A. Applicability of electrocoagulation process to the treatment of Ofloxacin and Chloramphenicol in aqueous media: Removal mechanism and antibacterial activity. Journal of Water Process Engineering. 2022. https://doi.org/10.1016/j.jwpe.2022.103080.
- Muttaqin R, Pratiwi R, Dewi ELR, Slamet MI. Degradation of methylene blue-ciprofloxacin and hydrogen production simultaneously using combination of electrocoagulation and photocatalytic process with Fe-TiNTAs, International Journal of Hydrogen Energy. International Journal of Hydrogen Energy. 2022. https://doi.org/10.1016/j.ijhydene.2022.04.031 .
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
