A efficient, regioselective and solvent free synthesis of 5,9-dihydropyrimido[4,5-e][1,2,4]triazolo[1,5-a]pyrimidine-6,8(4H,7H)-diones derivatives in presence nanocatalyst sulfuric acid grafted with silica-3-aminotriazol
Department of Chemistry, Yasouj University, PO. Box 353, Yasouj 75918-74831, Iran
Author and article information
Cite this as
Pourkazemi A, Karami B, Farahi M (2024) A efficient, regioselective and solvent free synthesis of 5,9-dihydropyrimido[4,5-e][1,2,4]triazolo[1,5-a]pyrimidine-6,8(4H,7H)-diones derivatives in presence nanocatalyst sulfuric acid grafted with silica-3-aminotriazol. Open J Chem. 2024; 10(1): 32-43. Available from: 10.17352/ojc.000037
Copyright License
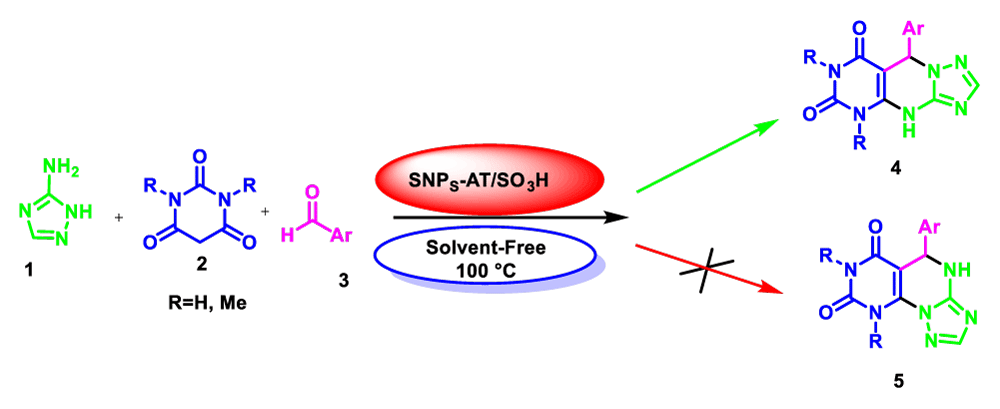
© 2024 Pourkazemi A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.In this research, SO3H-functionalized Silica grafted 3-amino triazol has been prepared and identified through FT-IR, XRD, EDX, Fe-SEM, and TGA spectra. 3-chloropropyltriethoxysilane was first reacted with silica nanoparticles and then treated with 3-amino-1H-1,2,4-triazoles and grafted finally with chlorosulfonic acid to prepare SiO2@(CH2)3-AT/SO3H (SNPS-AT/SO3H). Afterward, the catalytic efficiency has been investigated as a homogeneous and recyclable catalyst for the solvent-free synthesis of 5,9-dihydropyrimido[4,5-e][1,2,4]triazolo[1,5-a]pyrimidine-6,8(4H,7H)-diones by the one-pot multi-component reaction of aromatic aldehydes with 3-amino-1H-1,2,4-triazoles and 1,3-dimethyl barbituric acid under thermal (100 °C) conditions.
In recent times, nanoparticles have been investigated intensively due to the excellent attributes that produce them fitting for different applications including wetness sensors, pigments, drugstores, chemistry, and catalysts [1].
The creation of new and effective nanomaterials as catalyst supports is a challenge in new-fashioned research in catalysis that provides a good catalyst nanoparticle. Therefore, the use of them as heterogeneous supports has received increasing attention. Because they offer great surface areas on which both catalysis and interaction between the particles and their support material can occur [2].
Amidst various nanostructured substances, silica has maintained a pre-eminent position both in scientific studies and industrial design because of its facility of preparation and wide range of applications [3].
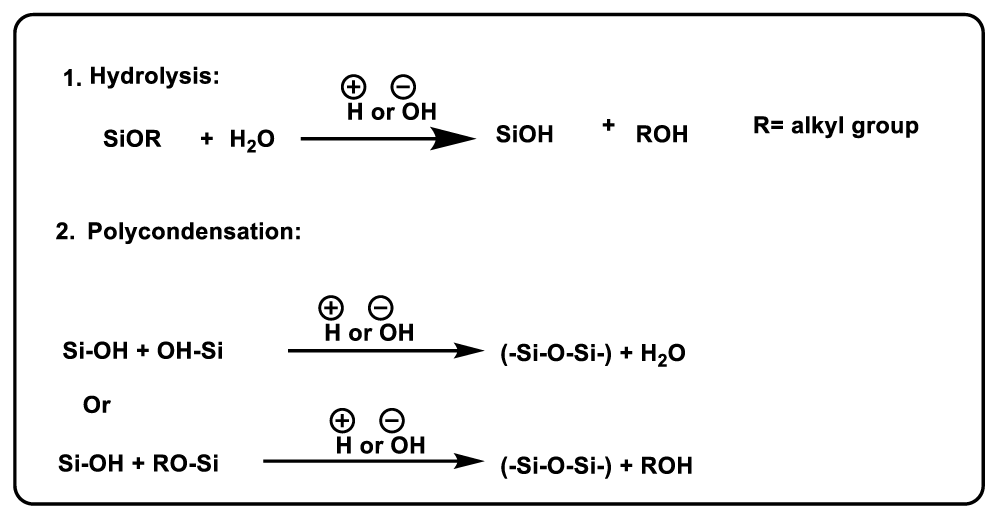
As the size distribution of silica nanoparticles is the fundamental criterion correlated to the quality of the terminal product, control of these agents is very essential. Recent research explicates that the sol-gel method has been confirmed to be a hopeful method for creating spherical silica nanoparticles, using tetraethylorthosilicate (TEOS) as a precursor due to its easiness and Inexpensive. This technique includes hydrolysis and polycondensation of the precursor in the presence of a base respectively. Which leads to the production of silanol groups that act as active reaction sites. This results in the formation of a cross-sectional three-dimensional grid structure and eventually siloxane (Si-O-Si) bridges, as explained in Scheme 1 [4].
Much research has been done on this method and it has been proven that the concentration of precursor, catalyst, co-solvent, water, and reaction temperature are among the parameters affecting the size and distribution of nanoparticles [4-6].
During the past several years, much notice has been paid to the manufacturing and use of acid catalysis in organic transformations. constantly, Homogeneous acid catalysts such as Chlorosulfonic acid, Hydrofluoric acid, Sulfuric acid, and Phosphoric acid catalytic and chemical processes received more attention from chemists [7-9]. However, the separation of these acid catalysts from the product and the need to neutralize acidic waste will limit their use. In this context, the use of nanoparticles as heterogeneous catalysts, reusable and strong solid acids in organic reactions, are often considered to follow the principles of green chemistry, which is catalyzed operations expend the least of energy and reagents and minimize waste [10,11].
The expansion and promotion of eco-friendly reactions is the most significant assignment in the simultaneous Scientists and chemical manufacturers. Therefore, due to the deficits of solvent used such as toxicity, high price, and difficulty of separation, performing chemical reactions in the condition absence of solvent has been highly regarded by researchers in industry and academia [12].
In chemistry, a multicomponent reaction (MCR), sometimes referred to as a “Multi-component assembly method” (MCAP), is a chemical reaction where more than two reactants combine in a sequential operation to give extremely selective products that retain the majority of the atoms of the starting material [13,14].
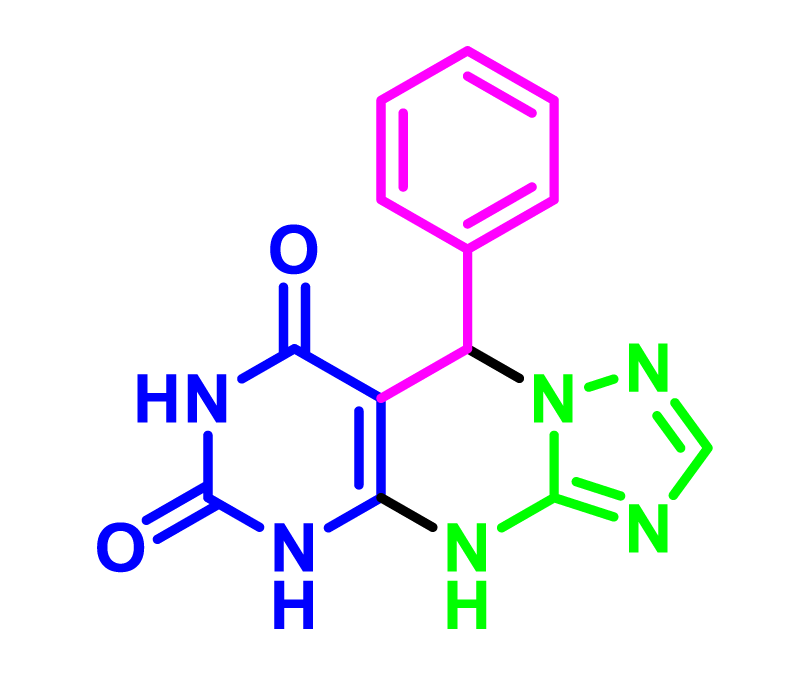
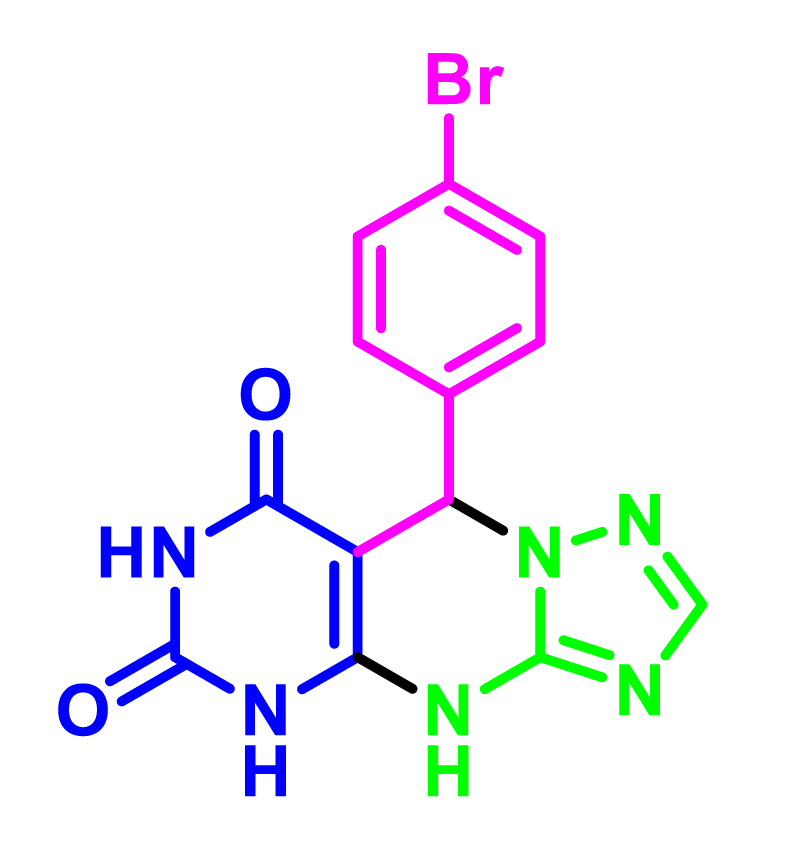
The synthesis of 5,9-dihydropyrimido [5,4- e][1,2,4]triazolo[1,5-a]pyrimidine-6,8(4H,7H)-diones derivatives is an attractive example of these MCRs. 1,2,4-Triazolopyrimidines are unique in the family of organic compounds because of their hopeful biological and pharmacological activities [15,16]. Research on these compounds has proven the medicinal properties of these compounds, such as anticancer, antiviral, antitumor, anti-inflammatory, antimicrobial, antifungal, antihistaminic, analgesic [17], anticonvulsive, and sedative [15] properties activities. Moreover, these compounds are used as cannabinoid receptor inverse agonists, anti-Alzheimer’s, anti-parasitic, and inhibitors of the bromodomain [18]. Accordingly, we have afforded a professional strategy for the synthesis of 1,2,4-Triazolopyrimidines derivatives utilizing accessible primitive materials, eco-friendly procedures, and a simple workup that has a great purpose.
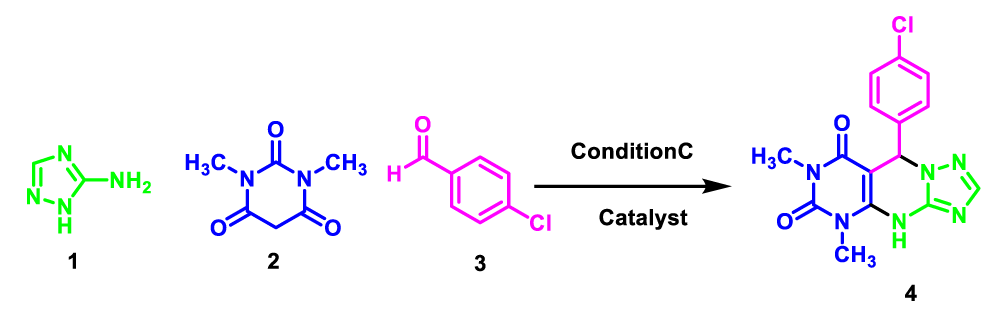
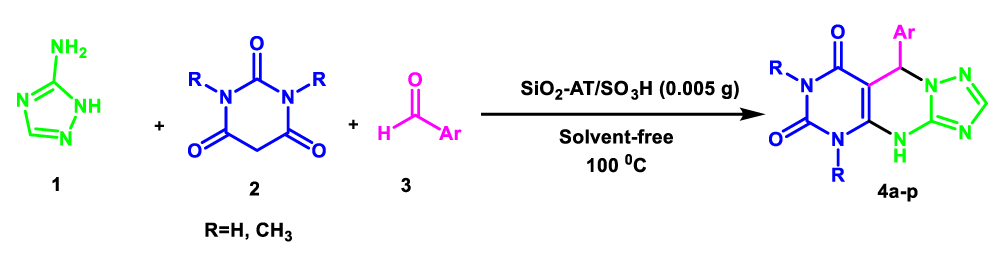
In view of the increasing public concern over environmental degradation and future resources, and in continuation of our previous reports in the field of heterogeneous catalysts [19-23]. In line with this research, silica nanoparticles were synthesized via sol-gel precipitation method, then 3-chloro propyltriethoxysilane was reacted with silica nanoparticles and treated with 3-amino-1H-1,2,4-triazoles and finally immobilization with chlorosulfonic acid as a novel heterogeneous catalyst for one-pot synthesis of 5,9-dihydropyrimido[e-4,5][1,2,4]triazolo[a-1,5]pyrimidine-6,8(4H,7H)-diones via three-component couplings of 3-amino-1H-1,2,4-triazoles (3-AT) 1, 1,3-dimethyl barbituric acid 2, and aldehydes 3 under solvent-free conditions (Scheme 2).
Experimental materials and equipment
All reagents and chemicals used including tetraethyl orthosilicate (TEOS), 3-chloropropyl tri methoxy silane (CPTMS), 3-amino-1H-1,2,4-triazoles(3-AT), Chlorosulfuric acid, potassium carbonate 1,3-dimethyl barbituric acid, and aldehydes, toluene, dichloromethane (DCM) and ethanol in this research were purchased from Fluka or Merck. The reaction process and the purity determination of the compounds were monitored by thin-layer chromatography (TLC) on commercial plates of silica gel SILG/UV254 (EtOAc: n-hexane, 1:3). In order to determine the identity and purity of the compounds, the melting point was determined by a measuring device of the German company Kruss. Fourier transform infrared (FTIR) spectra were recorded with potassium bromide pills and the FT-IR-680 plus spectrometer (Jasco, Japan) in the frequency range of 400-4000 cm-1. Another analysis used in determining the structure of the compounds synthesized in this study is Nuclear Magnetic Resonance. 1H NMR spectra in DMSO were registered on a BRUKER 400 MHz UltraShield spectrometer and 13C NMR spectra were recorded at 100 MHz using TMS as an internal standard (Bu Ali Research Institute, Mashhad, Iran). To determine the size and shape of the crystal, X-ray powder diffraction (XRD) patterns with Cu Kα radiation (λ= 0.15418 nm) model X’Pert Pro made in Germany, have been used. Energy dispersive spectroscopy (EDS) was achieved utilizing a TESCAN Vega model device. The morphology of the particles was examined via the Field Emission Scanning Electron Microscope (FE-SEM) under the acceleration voltage of 26 kV.
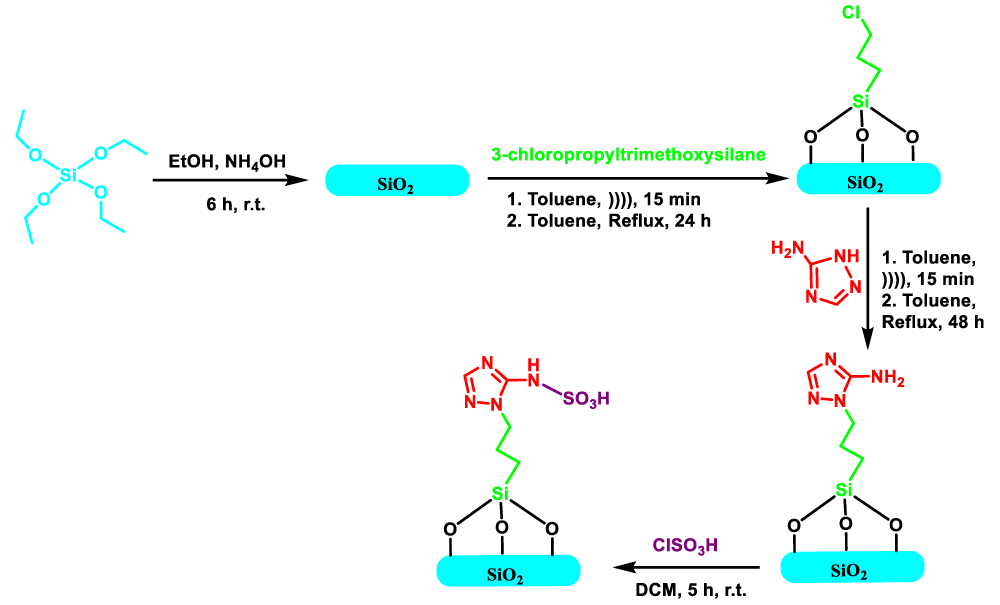
Synthesis of SiO2 nanoparticle
The SiO2 nanoparticles applied in this research, of the sol-gel procedure, due to their ability to form pure and homogenous results in gentle conditions, have been synthesized [24]. TEOS (6.2ml) was dispersed in ethanol (100 ml) and sonicated for 15 minutes to mix completely. Subsequently, ammonium hydroxide (6.5ml) as a catalyst was added to the mixture, under fast magnetic stirring for 6 h at room temperature. Then filtered by Centrifuges (4000 rpm, 30 min), to remove the solvent. finally washed (3x10) with an ethanol solution and water, and eventually, hydrophilic silica nanoparticles were obtained and dried at 60 °C for 12 h in a vacuum [25,26].
Connection CPTMS upon silica nanoparticles (SiO2 @ (CH2)3Cl)
Powder-white silica nanoparticle (1 g) was added to 60ml of dry toluene, and the reaction mixture was sonicated for 15 minutes to dissolve better. Subsequently, CPTMS (2 ml), dissolved in toluene, drop-drop into a rounded balloon was added and stirred under an argon atmosphere for 24 h. When the reaction mixture reaches room temperature, then filtered and washed with distilled water and ethanol. The resulting powder was dried in a vacuum at 80 C for 6 hours [7,19].
Grafted 3-AT with silica nanoparticles modified with (SiO2@(CH2)3-AT)
A mixture of the powder obtained from the previous step (SiO2 @ (CH2)3Cl) (1 g) and anhydrous toluene(30-mL) in a round-bottomed flask, for achieving a stable suspension, was dispersed by ultrasonic for 15 min. Then suspension of 3-AT (5 mmol) as a base, and K2CO3 (5 mmol), in dry toluene, was added to the reaction mixture under conditions (reflux, Ar atmosphere, time 48 h). Ultimately, the following off-white powder was filtered, then it was washed several times with (EtOH/ H2O) to remove surplus 3-AT, and SiO2@(CH2)3-AT dried at 100 °C in a Vacuum for 24 h.
Procedure for preparation of the SiO2@(CH2)3-AT/SO3H (SNPS-AT/SO3H).
In the flask containing SiO2@(CH2)3-AT (1 g), dried DCM (10 mL), and Et3N (0.2 mL) were added, and it was stirred to obtain a uniform solution. Subsequent 10 min, a solution of chlorosulfonic acid (1 mL) in dry DCM (3 mL) was slowly added drop-drop manner to the solution of SiO2@(CH2)3-AT at temperature 0 ˚C. Next addition was completed, and the flask contents were stirred at room temperature for 3-5 h. In the end, the residue, beige powder, is filtered and washed with DCM and ethanol before being dehydrated in an oven at 60 ◦C.
Catalyst titration for determining the acidity of SNPS-AT/SO3H
Acid-base titrations were employed to measure the acidity of the amount of acid stabilized on the surface of the SiO2@(CH2)3-AT, 100 mg of the catalyst was stirred in 20 ml double distilled water and titrated versus 0.1 N NaOH solution in the presence (2 drops) of phenolphthalein reagent. According to this investigation, the pH of the solution is 3.5. Also, the results resumed catalyst approval, with no substantial decrease in the amount of acid loading of the catalyst.
Assessment of the catalytic activity of SNPS-AT/SO3H by the synthesis of 1,2,4-Triazolo Pyrimidines
In brief, for the synthesis of 5,9-dihydropyrimido[5,4- e][1,2,4]triazolo[1,5-a]pyrimidine-6,8(4H,7H)-diones derivates: A mixture SNPS-AT/SO3H (0.005 g), 3-AT (1 mmol), 1,3-dimethyl barbituric acid (1 mmol), and aryl aldehyde (1 mmol) were stirred at 100 °C for 10 - 15 min in under solvent-free conditions. The rate of product formation was determined by TLC (EtOAc: n-hexane, 1:3). Subsequent completion of the reaction, the Balloon contents were cooled to room temperature, then warmed ethanol (5 mL) was added to the mixture, and the catalyst was collected by utilizing of Centrifuges instrument, and the powder was recrystallized from EtOH to yield the pure product.
Spectral data
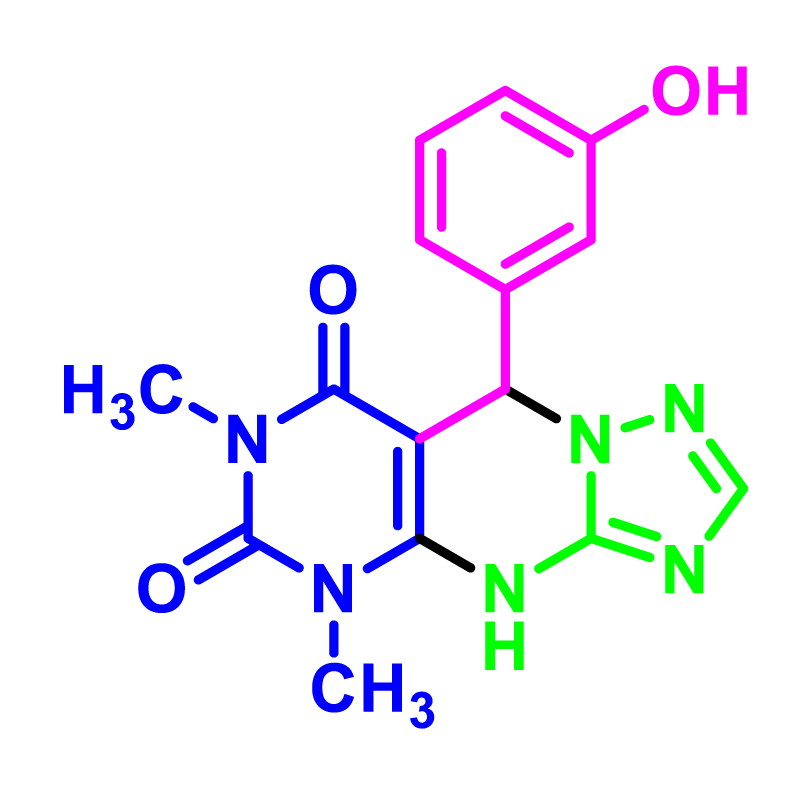
9-(3-hydroxyphenyl)-5,7-dimethyl-5,9-dihydropyrimido[4,5-e][1,2,4]triazolo[1,5-a]pyrimidine-6,8(4H,7H)-dione (4k). Light brown solid, M.p.: 210-213 °C, FT-IR (KBr, cm-1): 3380 (OH), 3262 (NH), 3012 (CH aromatic), 2958, 2914 (CH aliphatic), 1703, 1688 (C=O), 1599 (C=N), 1532, 1460 (C=C), 1388, 1267(C-N), 875 (Bending vibrations aromatic ring). 1H NMR (400 MHz, DMSO-d6) δ: 9.61 (s, 1H, NH), 9.47 (s, 1H, OH), 8.27 (s, 1H, Ar-H), 6.96-7.22 (m, 4H, Ar-H), 5.15 (s, 1H, CH), 2.92 (s, 3H, CH3), 2.80 (s, 3H, CH3). 13C NMR (100 MHz, DMSO-d6) δ: (162.7, 157.1) (C=O amin), 156.2 (C-OH), 151.9 (C-ring) (149.4, 148.6) (C- triazole ring), 138.1, 130.0, 120.0, 114.1, 111.9, 91.6 (C-Aromatice), 59.8 (C-benzyl), 29.1, 28.9 (CH3).
Results and discussion
The synthesis process of SNPS-AT/SO3H nanocatalyst is illustrated in Scheme 3. In the First Stage, SiO2 nanoparticles were fabricated through the sol-gel procedure. In the next step, the surface of silica nanoparticles was functionalized via (3-chloropropyl) trimethoxysilan molecule. Then, chloride the group was replaced by 3-AT, and finally SiO2@(CH2)3/AT nanostructure was sulfonated by chlorosulfonic acid to synthesize the desired nanocatalyst SNPS-AT/SO3H. The prepared nanocatalyst was characterized by FT-IR spectroscopy, EDX, XRD, FE-SEM, and TGA.
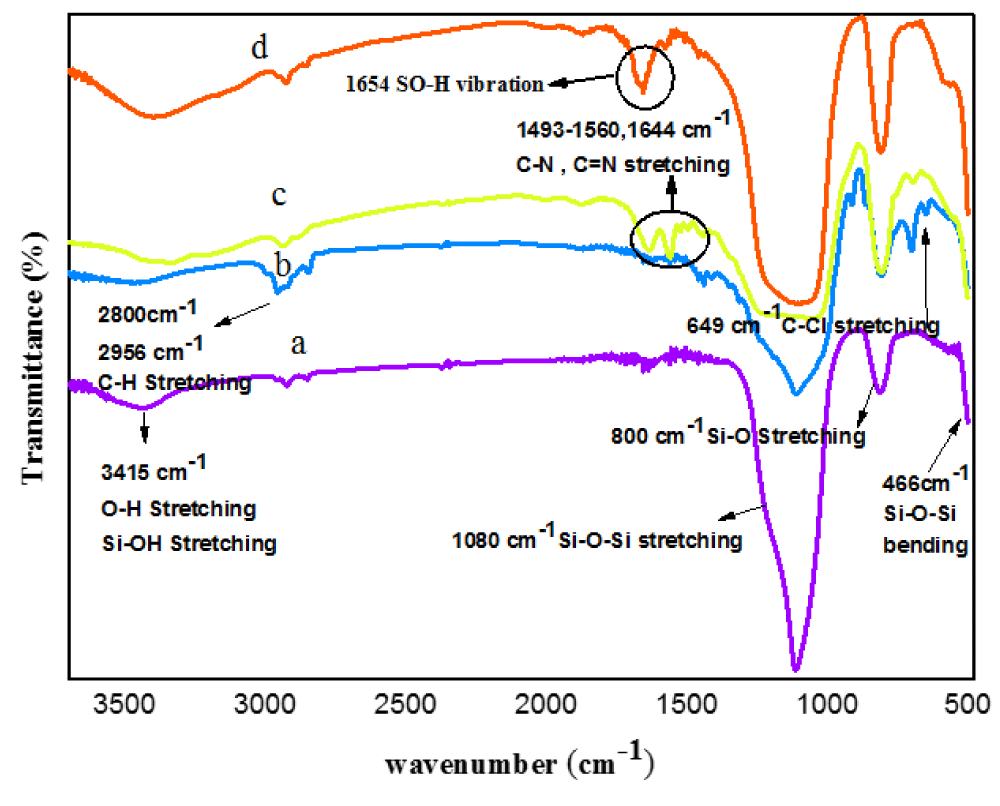
Figure 1 displays the FT-IR spectra of SiO2, SiO2@(CH2)3Cl, SiO2@(CH2)3/AT, and SNPS-AT/SO3H. In the FTIR spectrum of SiO2, a strong and evident peak at 1080 cm-1 can be related to the Si–O–Si symmetric stretching vibrations, also the absorption peaks at 800 and 466 cm-1 are due to the symmetric stretching vibration and bending vibration of Si-O, respectively. In addition, the absorption peak at 950 and 3415 cm-1 is attributed to the bending vibration of Si-OH and the vibrations of the hydroxyl group, respectively (Figure 1a) [27]. The absorption bands at 2800 - 2956 cm-1 correspond to the stretching vibration of a C–H band of CPTMS [28]. Furthermore, the absorption peak at 649cm-1 indicates the presence of the C-Cl bond [29] (Figure 1b). New peaks at 1493, 1560, and 1644 cm-1 may be assigned to C-N and C=N stretching, respectively [30,31]. Also, the intensity of the C-Cl peak decreased due to the presence of AT (Figure 1c). The successful sulfonation on the surface of the SiO2@(CH2)3/AT can be confirmed based on the vibrations at 1000 - 1300 cm-1 [32]. The stretching vibration of SOH bonds at 1070 cm-1 and the vibrational modes of N-SO2 bond at 1088 overlapped with a broad peak at 1080 -1262 those of strong Si-O-Si [33-36]. That attributed interaction of the sulfuric acid groups on the surface SiO2@(CH2)3/AT (Figure 1d). Accordingly, a comparison of all IR spectra confirms the successful preparation of the desired nano-catalyst SNPS-AT/SO3H
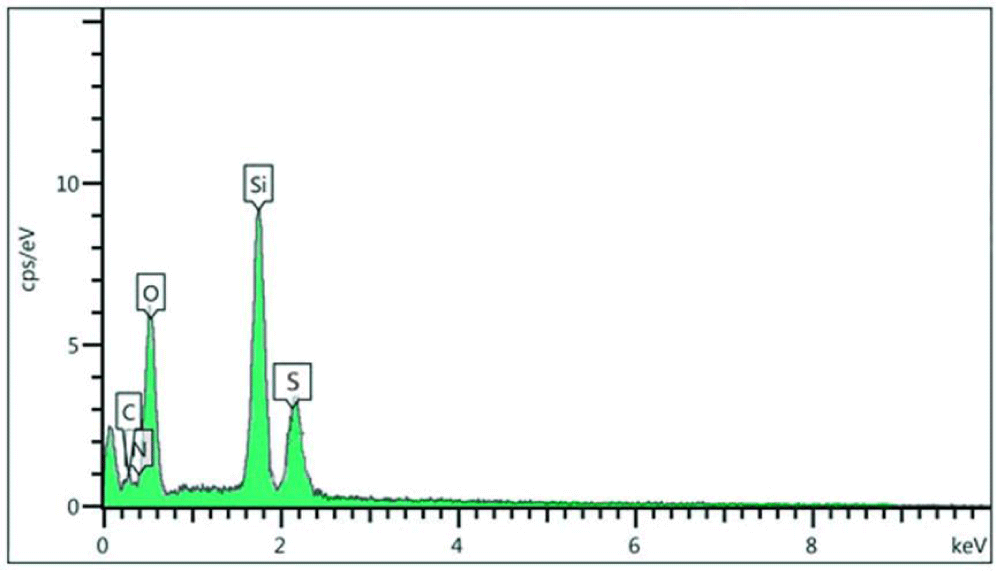
In addition, SNPS-AT/SO3H was characterized using Energy Dispersive X-ray Spectroscopy (EDS) analysis. This technique confirmed that the as-prepared catalyst was composed of Si, C, O, N, and S elements Figure 2.
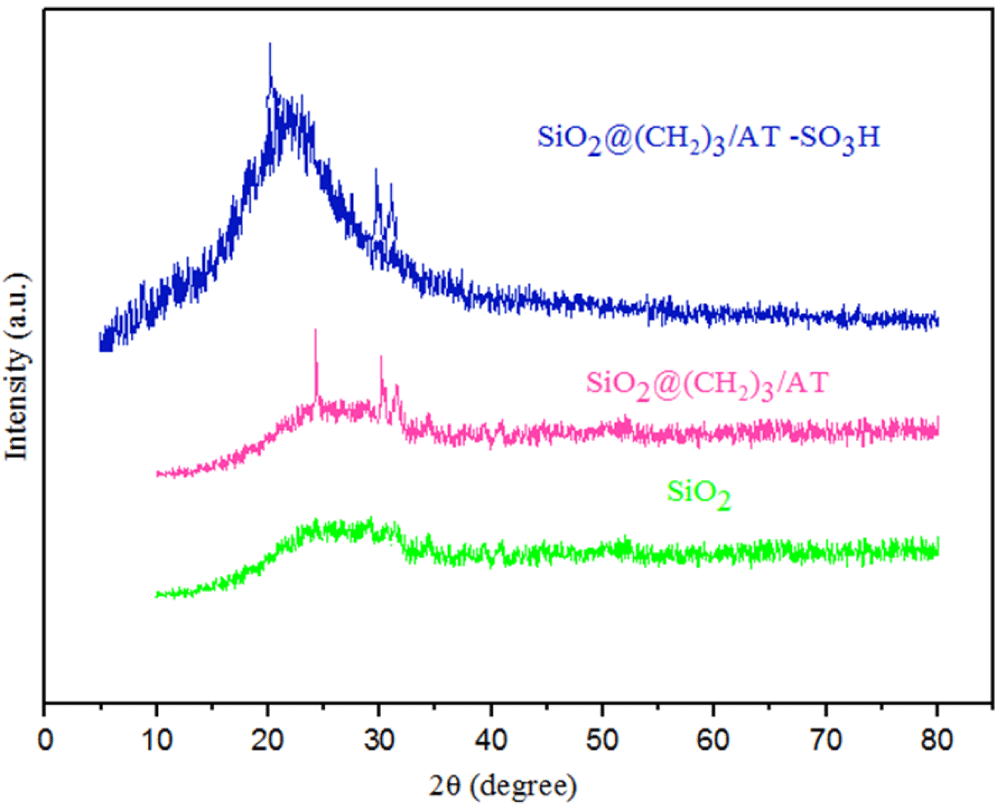
In order to verify the structure of the obtained nanocatalyst (SNPS-AT/SO3H), XRD analysis was performed and the resultant pattern of the prepared sample is displayed in Figure 3. As can be seen, a broad peak centered at 2θ = 24. shows which is consistent with an amorphous nature of SiO2 structure as reported in the literature [19]. The XRD pattern showed the nanoparticles SiO2@(CH2)3/AT and in comparison with SiO2, the existence of nitrogen in the structure confirmed the formation of SiO2@(CH2)3/AT. The other present groups in the catalyst did not show any changes in the nanocatalyst structure.
In another assay, the size and morphology of the SNPS-AT/SO3H were recognized using FE-SEM. Based on these results, the size of the as-prepared catalyst particles size is about 20 nm, and the nanocatalyst (SNPS-AT/SO3H) particles are in the nanometer scale and have a nearly spherical shape (Figure 4).
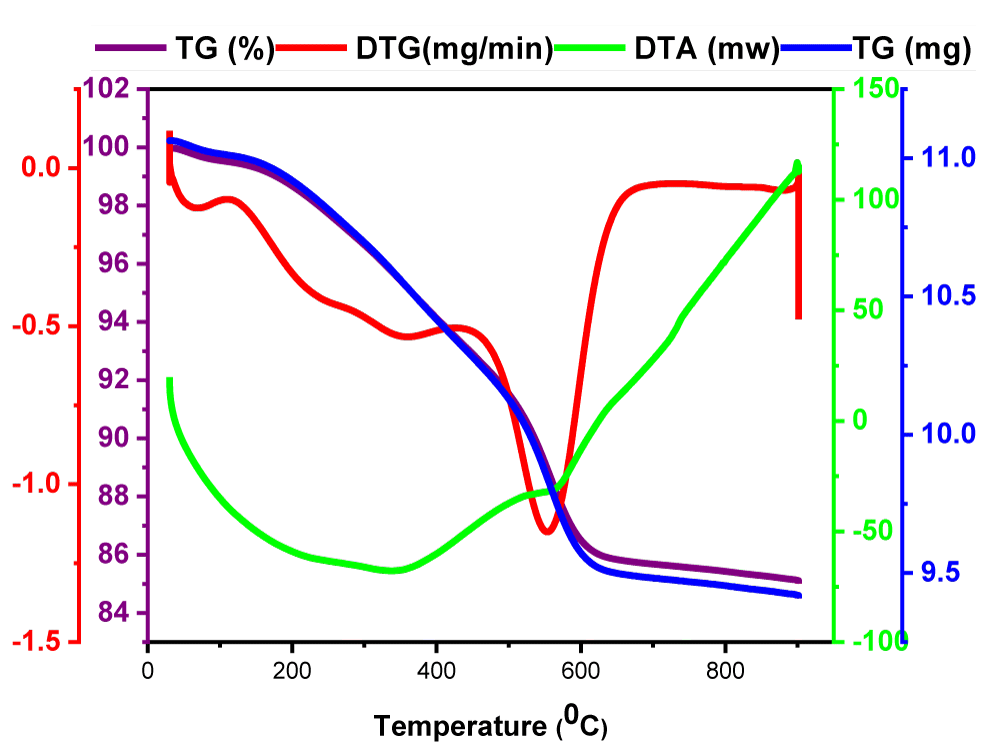
Thermogravimetric analysis (TG) of the SiO2-AT/SO3H shows in (Figure 5) the thermal stability of the as-prepared sample. The results showed a weight loss (about 4%) at temperatures below 100 degrees °C corresponding to the elimination of H2O and other organic solvents; however, the second weight loss observed in the temperature range of 170 to 280 °C (about 4%) attributed to the organic components located on the surface of the NPs. Also, The weight loss related to sulfonic acid groups (about 2%) was observed at 300 °C - 380 °C. The largest weight loss (about 8%) can be assigned to the thermal decomposition of the silica NPs at 480 °C - 550 °C. The thermal decomposition of the differential Thermogravimetric analysis (DTG) shows that the decomposition of the organic structure is mainly at a temperature of 550 ° C, and the catalyst is quite stable up to a temperature of 200 ° C. Therefore, the stability of the synthesized nanocatalyst was confirmed [19,32].
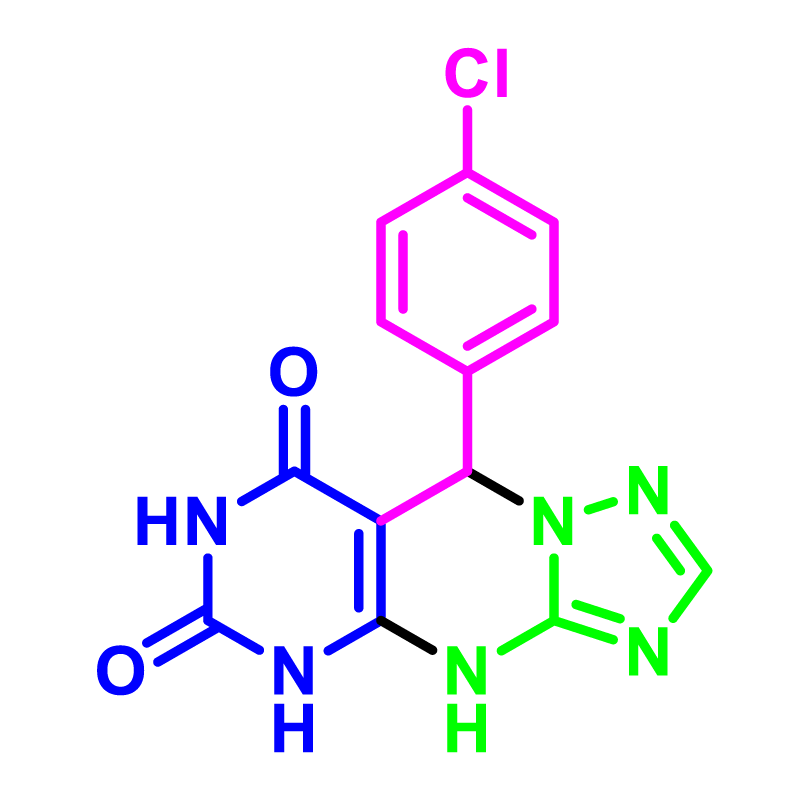
The catalytic activity of the SNPS-AT/SO3H was investigated for the synthesize 5,9-dihydropyrimido[5,4- e][1,2,4]triazolo[1,5-a]pyrimidine-6,8(4H,7H)-diones. In order to optimize the reaction conditions, we selected 3-AT, 1,3-dimethyl barbituric acid, and 4-choloro benzaldehyde as the modal reaction. We used several protic, aprotic solvents and solvent-free conditions in the presence of various amounts of the SNPS-AT/SO3H catalyst in the modal reaction. Results Table 1 shows that solvent-free conditions obtain better yields than aprotic and protic solvents. Also, the influence of temperature was investigated for this reaction and it was found that the reaction would yield further at 100 °C temperature. Finally, the thermal-assisted model reaction is efficiently carried out by adding catalytic amounts of NPs-AT/SO3H (0.005 g) in a solvent-free condition.
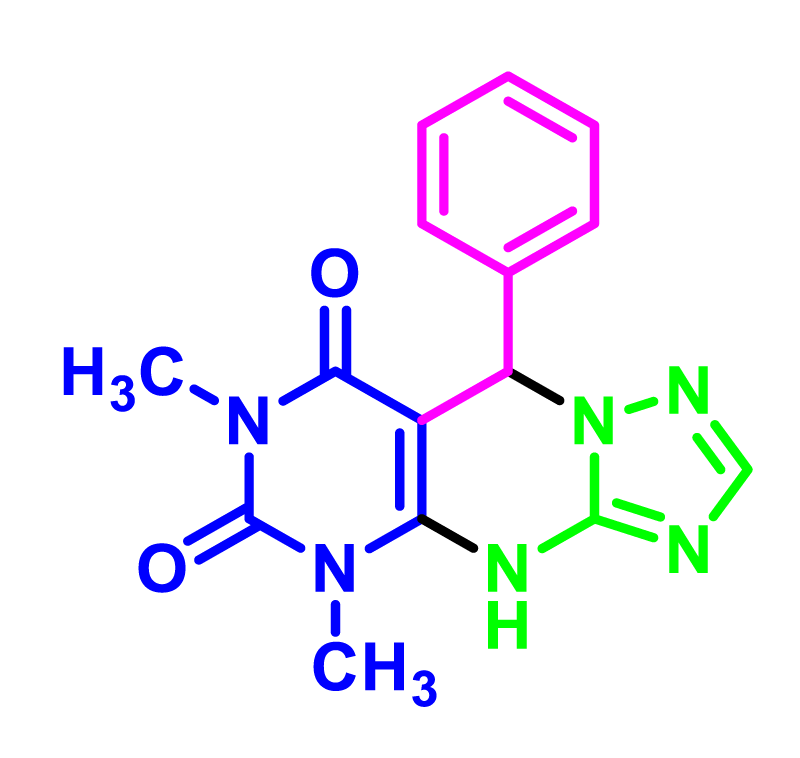
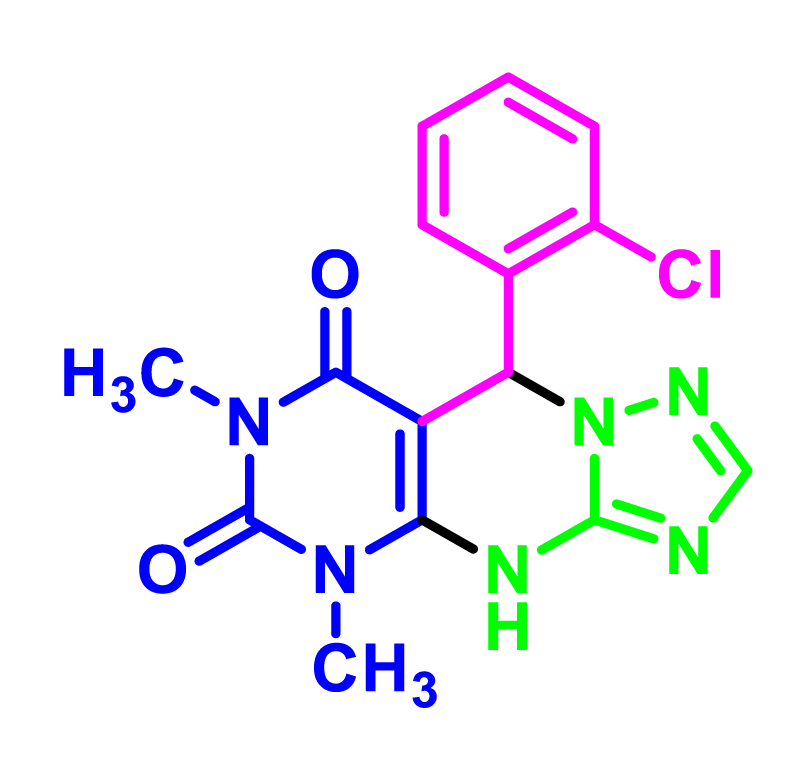
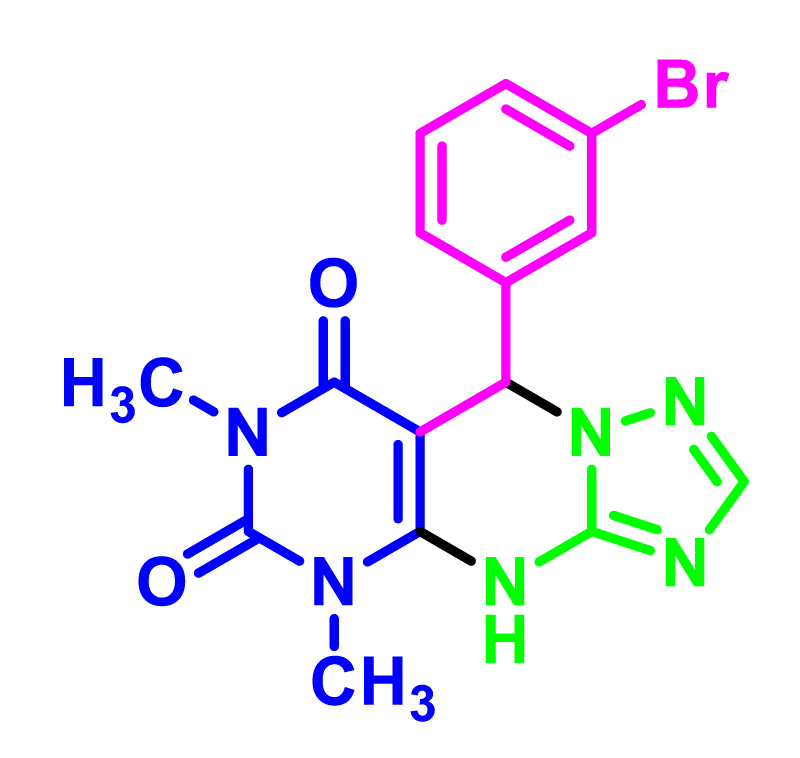
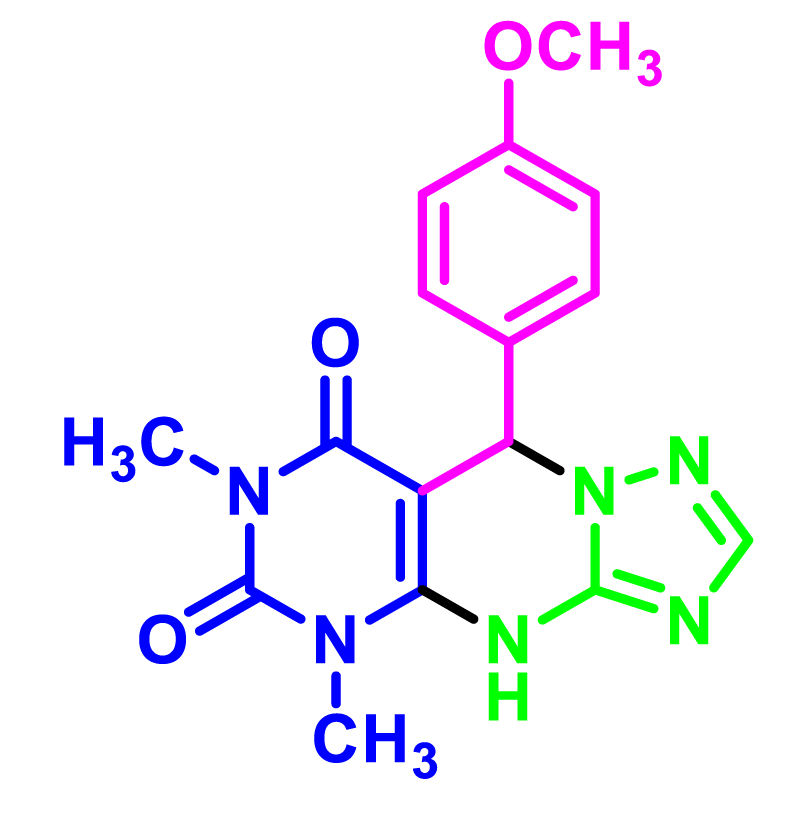
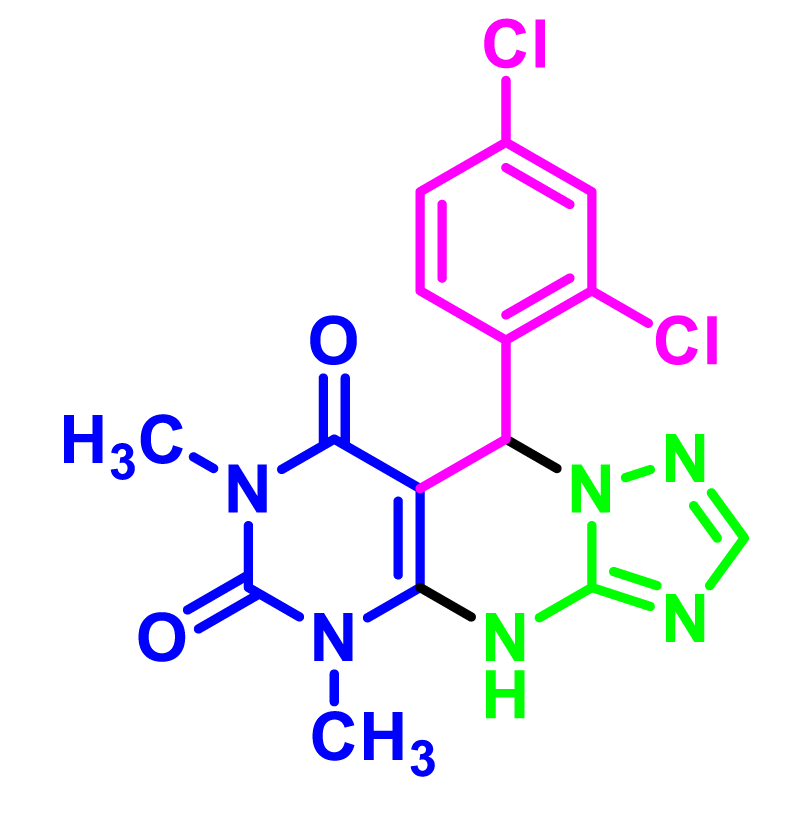
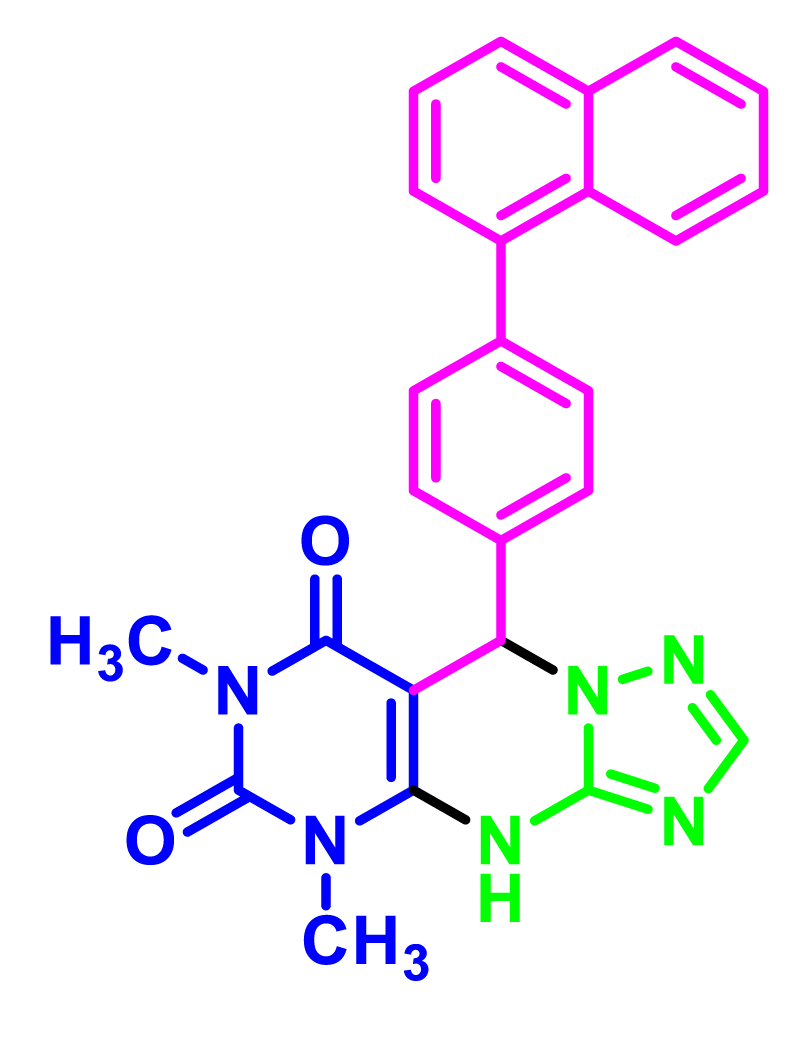
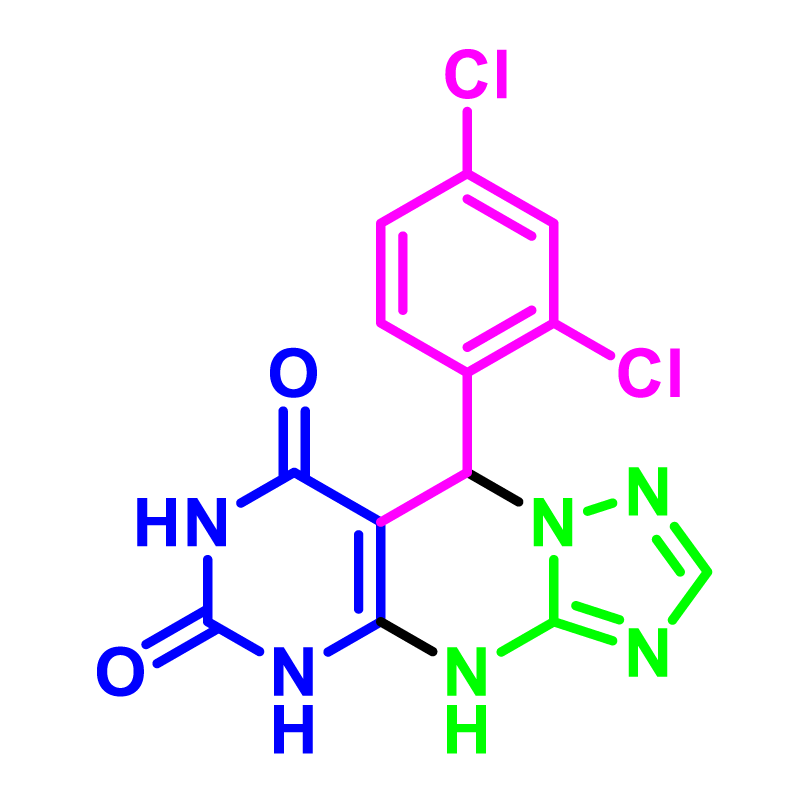
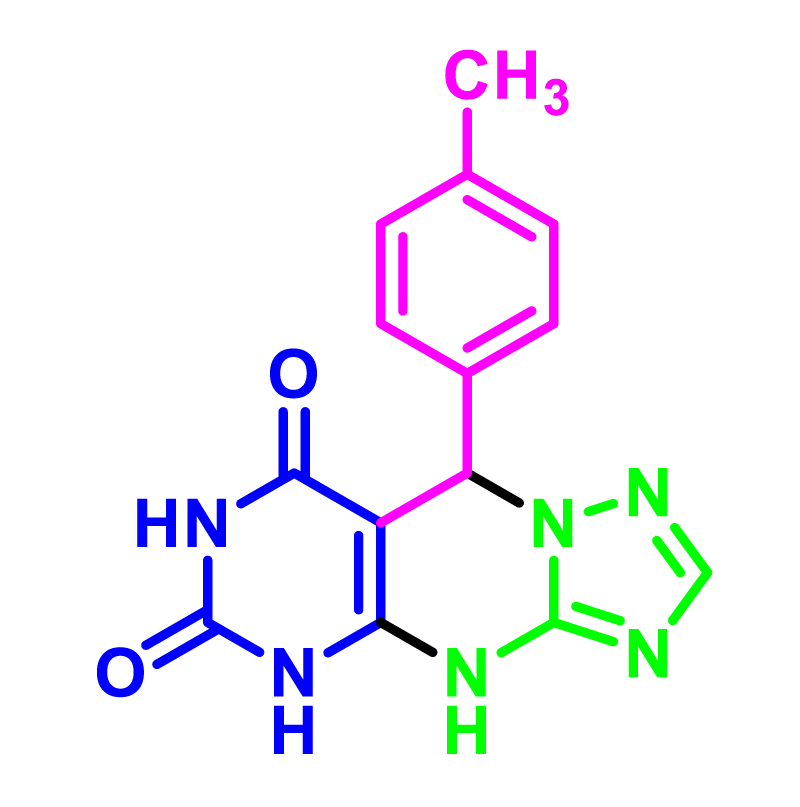
In order to develop the scope of this reaction, a wide range of both electron-rich and electron-poor aryl aldehydes were used. It was located that yield electron-rich more than electron-poor aryl aldehyde reacted well in this process. All the corresponding products were obtained with excellent yields. The products were isolated and confirmed by a comparison of their physical data and spectroscopic techniques with those reported in the literature. The results are summarized in Table 2 [15,18,24].
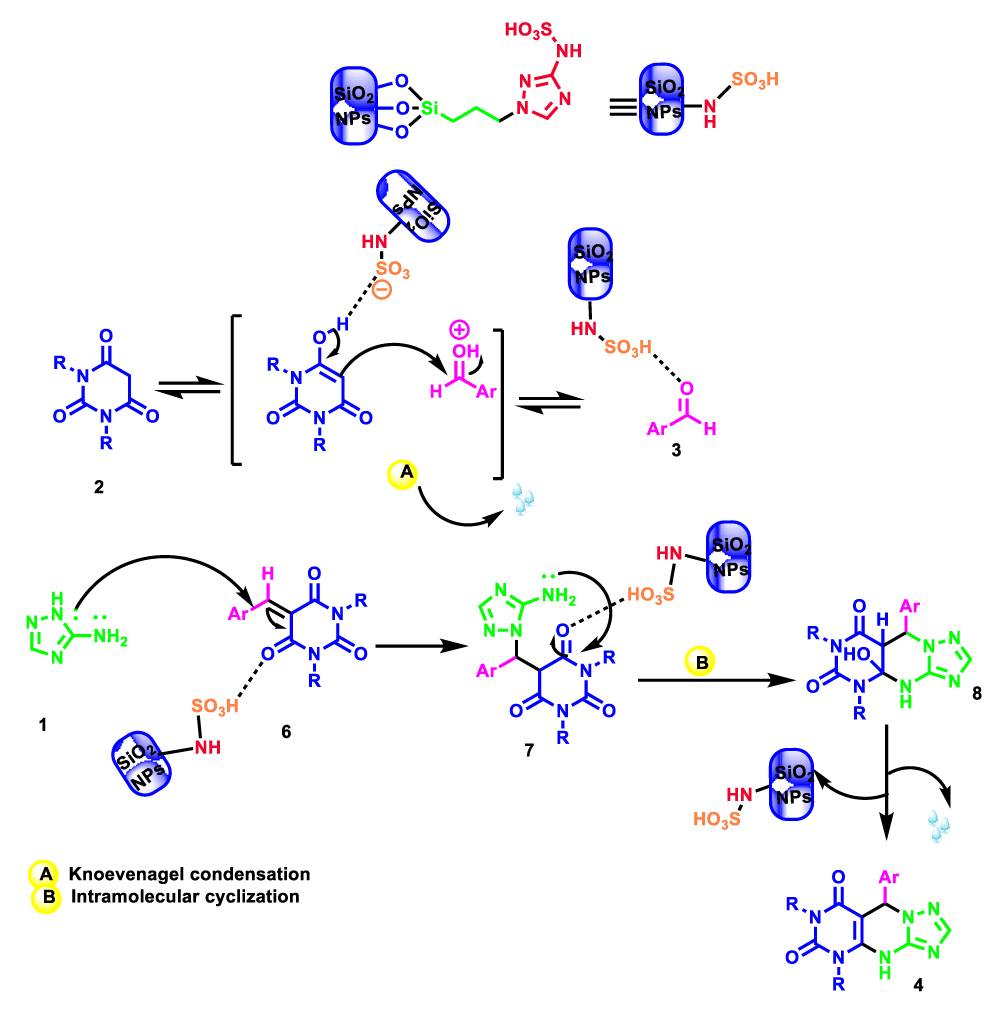
1H NMR and 13C NMR spectra proved the suggested products. 9-aryl-5,9-dihydropyri-mido[4,5-d][1,2,4]triazolo[1,5-a]pyrimidine-6,8(4H,7H)-diones 4 was formed of the reaction aromatic aldehydes 1, dimethyl barbituric acid 2 and 3-amino-1H-1,2,4-triazoles 3 in the presence of nanocatalyst SNPS-AT/SO3H and in the absence of solvent. Generally, both regioisomers 4 and 5 are possible reactions, but in this reaction, the alternative product 5 wasn’t formed. 1H NMR spectroscopic analysis confirms this claim.
The suggested mechanism for the formation of 5,9-dihydropyrimido[5,4-e][1,2,4]triazolo[1,5-a]pyrimidine-6,8(4H,7H)-diones are shown in Scheme 4. Initially, 1,3-diethyl barbituric 2 and aryl aldehyde 3 are activated by SNPS-AT/SO3H. Then arylidene-1,3-diethyl barbituric acid 6 is achieved from the Knoevenagel condensation reaction between 1,3-diethyl barbituric acid with aryl aldehyde. A Michael-type addition between Compound 6 and 3-amino-1H-1,2,4-triazole 1 gives the intermediate 7, and subsequent heterocyclization and dehydration give the corresponding products 4. Scheme 4. A plausible mechanism for the formation of 5,9-dihydropyrimido[5,4-e][1,2,4]triazolo[1,5-a]pyrimidine-6,8(4H,7H)-diones 4 catalyzed by SNPS-AT/SO3H.
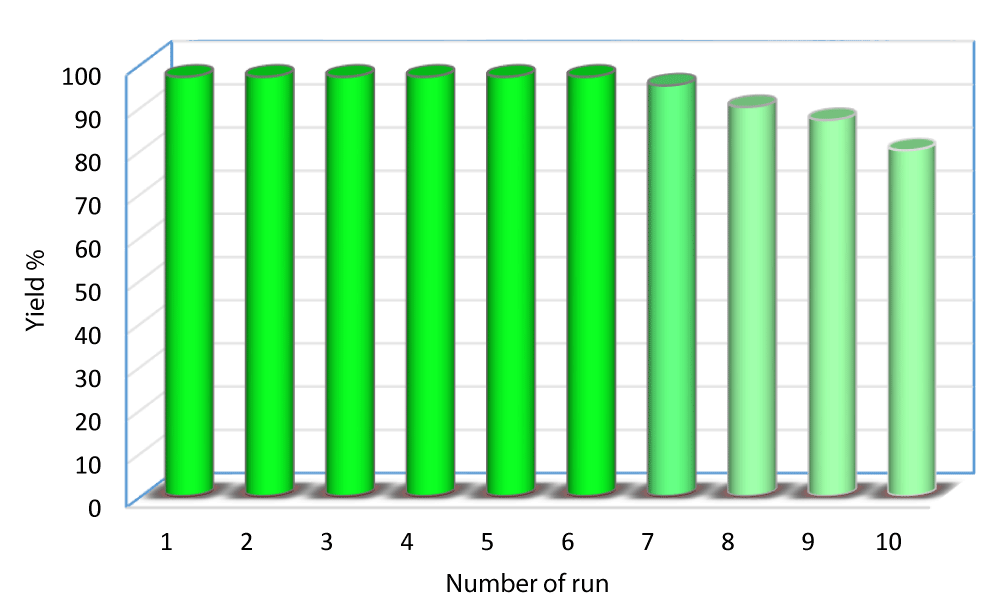
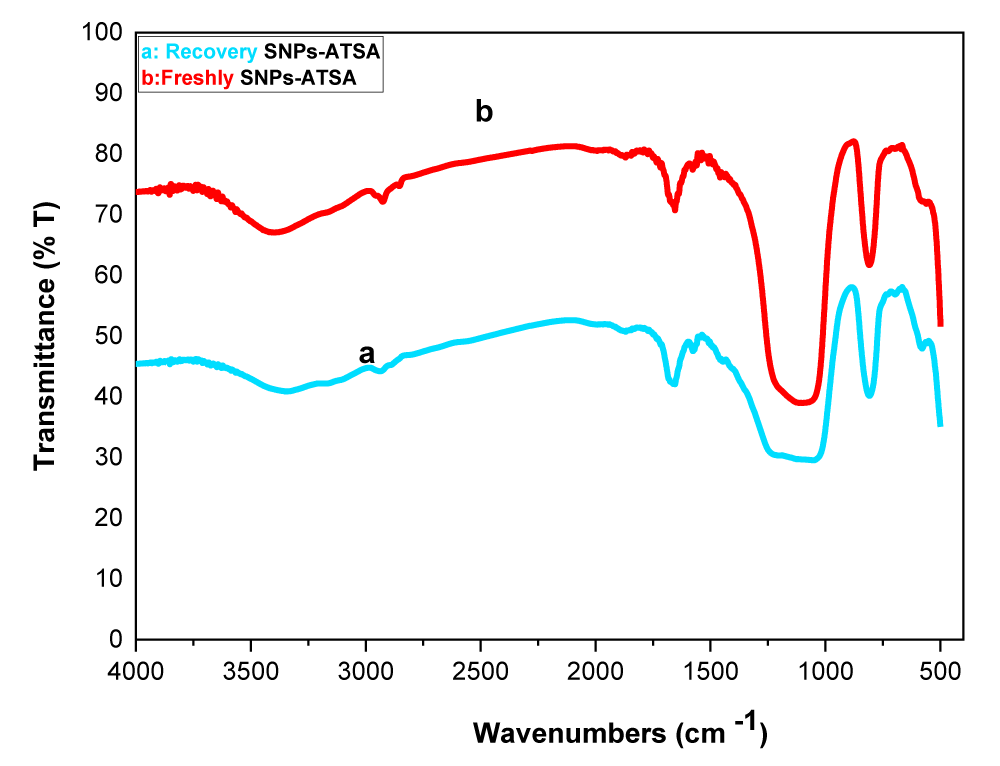
SNPS-AT/SO3H (0.005 g) was added to a mixture of 3-AT (1 mmol), 1,3-dimethyl barbituric acid (1 mmol), and 4-chlorobenzaldehyde (1 mmol) in optimal conditions. After completing the reaction, ethyl acetate (5 ml) was added to the reaction mixture SNPS-AT/SO3H catalyst was removed from the product obtained using a filtering. The catalyst was washed accurately with EtOH: H2O (3×10) and dried at 100 °C in a vacuum oven overnight. Using this method, the catalyst recovered is reused up to 6 times with preservation of performance, and only activity decreases insignificantly after six catalytic cycles (Figure 6). The FT-IR spectra of the catalyst after six cycles (a) have shown the same spectral fingerprint of the newly prepared catalyst (b) designating recycled catalyst chemical stability (Figure 7).
Conclusion
In conclusion, a new nanocatalyst [SNPS-AT/SO3H] was successfully synthesized and characterized for the formation of 5,9-dihydropyrimido[5,4-e][1,2,4]triazolo[1,5-a]pyrimidine-6,8(4H,7H)-diones. The efficiency of this catalysis under solvent-free conditions has made this method green, and environmentally friendly. Also, The catalyst showed an excellent yield in a short time. Another advantage of this catalyst is easy separation from the reaction mixture and reuse without any considerable loss in catalytic activity.
The authors gratefully acknowledge partial support of this work by Yasouj University, Iran.
- Rao KS, El-Hami K, Kodaki T, Matsushige K, Makino K. A novel method for synthesis of silica nanoparticles. J Colloid Interface Sci. 2005 Sep 1;289(1):125-31. doi: 10.1016/j.jcis.2005.02.019. PMID: 15913636.
- Veisi H, Azizi S, Mohammadi P, Green synthesis of the silver nanoparticles mediated by Thymbra spicata extract and its application as a heterogeneous and recyclable nanocatalyst for catalytic reduction of a variety of dyes in water. J Clean Prod. 2018; 170: 1536–1543.
- Dabbaghian MA, Babalou AA, Hadi P, Jannatdoust E, A parametric study of the synthesis of silica nanoparticles via sol-gel precipitation method. Int J Nanosci Nanotechnol. 2010; 6: 104–113.
- Sharma RK, Sharma S, Dutta S, Zboril R, Gawande MB, Silica-nanosphere-based organic-inorganic hybrid nanomaterials: synthesis, functionalization and applications in catalysis. Green Chem 2015; 6: 3207-3230.
- Azlina H, Hasnidawani J, Norita H, Surip S, Synthesis of SiO₂ Nanostructures Using Sol-Gel Method. Acta Phys Pol A. 2016; 29: 842–844.
- Ambrogio MW, Frasconi M, Yilmaz MD, Chen X. New methods for improved characterization of silica nanoparticle-based drug delivery systems. Langmuir. 2013 Dec 10;29(49):15386-93. doi: 10.1021/la402493q. Epub 2013 Nov 25. PMID: 24199945.
- Akrami S, Karami B, Farahi M, Preparation and characterization of novel phthalhydrazide-functionalized MCM-41 and its application in the one-pot synthesis of coumarin-fused triazolopyrimidines. RSC Adv 2017; 7: 34315–34320.
- Khosravian F, Karami B, Farahi M, Synthesis and characterization of molybdic acid immobilized on modified magnetic nanoparticles as a new and recyclable catalyst for the synthesis of chromeno [4, 3-b] chromenes. New J Chem. 2017; 41: 11584–11590.
- Cole-Hamilton DJ. Homogeneous catalysis--new approaches to catalyst separation, recovery, and recycling. Science. 2003 Mar 14;299(5613):1702-6. doi: 10.1126/science.1081881. PMID: 12637737.
- Safari J, Zarnegar Z, A magnetic nanoparticle-supported sulfuric acid as a highly efficient and reusable catalyst for rapid synthesis of amidoalkyl naphthols. J Mol Catal A Chem. 2013; 379,: 269–276.
- Keyhaniyan M, Shiri A, Eshghi H, Khojastehnezhad A, Novel design of recyclable copper (II) complex supported on magnetic nanoparticles as active catalyst for Beckmann rearrangement in poly (ethylene glycol). Appl Organomet Chem. 2018; 32: 1–10
- Ghorbani-Vaghei R, Karimi-Nami R, Toghraei-Semiromi Z, Amiri M, Ghavidel M, One-pot synthesis of aliphatic and aromatic 2H-indazolo [2, 1-b] phthalazine-triones catalyzed by N-halosulfonamides under solvent-free conditions. Tetrahedron. 2011; 67: 1930–1937.
- Brahmachari G, Banerjee, B Facile and one-pot access to diverse and densely functionalized 2-amino-3-cyano-4 H-pyrans and pyran-annulated heterocyclic scaffolds via an eco-friendly multicomponent reaction at room temperature using urea as a novel organo-catalyst. ACS Sustain Chem Eng. 2014; 2: 2802–2812.
- Karami B, Farahi M, Akrami S, Elhamifar D, Tungstic acid-functionalized MCM-41 as a novel mesoporous solid acid catalyst for the one-pot synthesis of new pyrrolo [2, 1-a] isoquinolines. New J Chem. 2018; 42: 12811–12816.
- Farahi M, Karami B, Banaki Z, Improved microwave-assisted catalyst-free synthesis of 9-aryl-5, 9-dihydropyrimido [4, 5-d][1, 2, 4] triazolo [1, 5-a] pyrimidine-6, 8 (4H, 7H)-dione derivatives. Chinese Chem Lett. 2015; 26: 1065–1067.
- Gladkov ES, Gura KA, Sirko SM, Desenko SM, Groth U, Chebanov VA. Features of the behavior of 4-amino-5-carboxamido-1,2,3-triazole in multicomponent heterocyclizations with carbonyl compounds. Beilstein J Org Chem. 2012;8:2100-5. doi: 10.3762/bjoc.8.236. Epub 2012 Nov 30. PMID: 23243471; PMCID: PMC3520566.
- Karami B, FarahibM, Banaki Z, A new protocol for catalyst-free regioselective synthesis of 5, 9-dihydropyrimido [5, 4-e][1, 2, 4] triazolo [1, 5-a] pyrimidine-6, 8 (4H, 7H)-diones. Synlett. 2015; 26: 741–744.
- Abdollahi-Basir MH, Shirini F, Tajik H, Ghasemzadeh MA, A facile and regioselective synthesis of some new pyrimido [4, 5-d][1, 2, 4] triazolo [1, 5-a] pyrimidinediones catalyzed by Zn (BDC)-MOF under ultrasound irradiation J Mol Struct. 2019; 1195: 302–308.
- Pourkazemi A, Asaadi N, Farahi M, Zarnegaryan A, Karami B, Glucose-Decorated Silica-Molybdate Complex: A Novel Catalyst for Facile Synthesis of Pyrano [2, 3-d]-Pyrimidine Derivatives. Acta Chim Slov. 2022; 69: 1-9.
- Gholtash J E, Farahi M, Tungstic acid-functionalized Fe3 O4@ TiO2: preparation, characterization and its application for the synthesis of pyrano [2, 3-c] pyrazole derivatives as a reusable magnetic nanocatalyst. RSC Adv Pap. 2018; 8: 40962–40967.
- Farahi M, Karami B, Sedighimehr I, Tanuraghaj HM, An environmentally friendly synthesis of 1, 4-dihydropyrano [2, 3-c] pyrazole derivatives catalyzed by tungstate sulfuric acid. Chinese Chem Lett. 2014; 25:1580–1582.
- Keshavarz R, Farahi M. Novel cellulose supported 1,2-bis(4-aminophenylthio)ethane Ni(ii) complex (NiII(BAPTE)(NO3)2-Cell) as an efficient nanocatalyst for the synthesis of spirooxindole derivatives. RSC Adv. 2022 Jan 27;12(6):3584-3592. doi: 10.1039/d1ra08182a. PMID: 35425356; PMCID: PMC8979259.
- Rohani L, Karami B, Farahi M, A green, facile, and one‐pot synthesis of 2‐amino‐4 H‐pyran derivatives using tungstate sulfuric acid as a solid acid catalyst. J Chinese Chem Soc. 2021; 68: 888–892.
- Karami B, Farahi M, Banaki Z, A new protocol for catalyst-free regioselective synthesis of 5, 9-dihydropyrimido [5, 4-e][1, 2, 4] triazolo [1, 5-a] pyrimidine-6, 8 (4H, 7H)-diones. Synlett. 2015; 26: 741–744.
- Rahman IA, Padavettan V, synthesis of silica nanoparticles by sol-gel: size-dependent properties, surface modification, and applications in silica-polymer nanocomposites—a review. J Nanomater. 2012; 2012: 1-15
- Green DL, Lin JS, Lam YF, Hu MZ, Schaefer DW, Harris MT. Size, volume fraction, and nucleation of Stober silica nanoparticles. J Colloid Interface Sci. 2003 Oct 15;266(2):346-58. doi: 10.1016/s0021-9797(03)00610-6. PMID: 14527458.
- Jiang X, Tang X, Tang L, Zhang B, Mao H, Synthesis and formation mechanism of amorphous silica particles via sol-gel process with tetraethylorthosilicate. Ceram Int. 2019; 45: 7673–7680.
- Akrami S, Karami B, Farahi M, Preparation and characterization of novel phthalhydrazide-functionalized MCM-41 and its application in the one-pot synthesis of coumarin-fused triazolopyrimidines. RSC Adv. 2017; 41: 34315–34320.
- Ariannezhad M, HabibiD, Heydari S, Copper nanoparticles: A capable and versatile catalyst for the synthesis of diverse 1-phenyl-1H-tetrazoles from amino acids. Polyhedron. 2019; 160:170-179.
- Veisi H, Taheri S, Hemmati S, Preparation of polydopamine sulfamic acid-functionalized magnetic Fe3O4 nanoparticles with a core/shell nanostructure as heterogeneous and recyclable nanocatalysts for the acetylation of alcohols, phenols, amines and thiols under solvent-free conditions. Green Chem. 2016; 18: 6337–6348.
- Tayebee R, Amini MM, Pouyamanesh S, Aliakbari A, A new inorganic-organic hybrid material Al-SBA-15-TPI/H6P2 W18O62 catalyzed one-pot, three-component synthesis of 2 H-indazolo [2, 1-b] phthalazine-triones. Dalt Trans. 2015; 44: 5888–5897.
- Barzkar A, Beni AS. In situ synthesis of SO 3 H supported Fe3O4@resorcinol-formaldehyde resin core/shell and its catalytic evaluation towards the synthesis of hexahydroquinoline derivatives in green conditions. RSC Adv. 2020 Nov 13;10(68):41703-41712. doi: 10.1039/d0ra06972h. PMID: 35516541; PMCID: PMC9057767.
- Pourkazemi A, Zare A, Characterization and Application of SO 3 H-functionalized Phthalimide (SFP) as an Efficient and Recyclable Catalyst for the Solvent-free Synthesis of 2-Amino-4H-chromenes. Org Chem Res. 2016; 2: 88-95.
- Farahi M, Karami B, Keshavarz R, Khosravian F, Nano-Fe3O4@ SiO 2 -supported boron sulfonic acid as a novel magnetically heterogeneous catalyst for the synthesis of pyrano coumarins. RSC Adv. 2017; 7: 46644–46650.
- Nasresfahani Z, Kassaee MZ, Eidi E, Efficient synthesis of α-aminonitriles over homopiperazine sulfamic acid functionalized mesoporous silica nanoparticles (MSNs-HPZ-SO 3 H), as a reusable acid catalyst. J Iran Chem Soc. 2019; 16: 1819–1825.
- Zhou S, Long M, Wu L, Lei M, Bai J, Huang K, Yin D, Titanate nanotubes covalently bonded sulfamic acid as a heterogeneous catalyst for highly efficient conversion of levulinic acid into n-butyl levulinate biofuels. Biomass Convers Biorefinery. 2022; 1-13.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.



 Save to Mendeley
Save to Mendeley
